-
非小细胞肺癌(non-small cell lung cancer, NSCLC)是肺癌中最常见的组织学类型,发病率逐年增高,无论是在我国还是全球,都已成为致死率最高的癌症之一[1]。因此,对于NSCLC的防治,特别是探索新的化学预防策略显得尤为重要。目前新兴出现的以靶向和免疫疗法为手段治疗NSCLC越来越引人关注,尽管如此,对于肺癌的治疗同样也面临着研发新型抗癌药物的挑战,包括研发过程中所需巨额费用和时间,而重新使用具有潜在癌症治疗的非抗癌药物用于癌症治疗,不仅可以节省研发新药所需的人、财、物力,甚至还可能提高治疗效果。临床前研究和数据证实许多非抗癌药物具有潜在的预防和治疗癌症的作用,譬如对NSCLC的预防和治疗,这些药物就有阿司匹林、他汀类药物等[2]。
阿司匹林和阿托伐他汀是广泛用于心血管疾病的一线防治用药。近年来,它们已经被证实可能成为新兴的、可用于不同类型肿瘤(包括NSCLC)的化学预防药物[3-5]。尽管它们抗NSCLC的确切分子机制尚需要进一步研究,但文献报道其抗肿瘤作用机制存在重叠,即均可靶向作用于mTOR和NFκB通路。据此我们假设:阿司匹林和阿托伐他汀联合使用可通过共同抑制mTOR和NFκB通路而发挥协同抗NSCLC效应。为验证假设,本实验选取了非小细胞肺癌A549和NCI-H460细胞,在体外研究阿司匹林联合阿托伐他汀对它们细胞增殖的影响及其相关作用机制,期望在应用阿司匹林和阿托伐他汀防治心血管疾病的同时可以降低NSCLC的患病风险,以及为设计新型控制NSCLC的干预策略提供理论依据。
HTML
-
人非小细胞肺癌A549和NCI-H460细胞(美国Manassas公司),培养于含10%胎牛血清的DMEM培养基(美国Sigma-Aldrich公司),置37 ℃饱和湿度、5% CO2培养箱孵育。阿司匹林和阿托伐他汀标准品化合物(美国Sigma-Aldrich公司),用DMSO溶解后−20 ℃保存。RNA提取试剂盒购自上海博彩生物科技公司,BCA法蛋白定量试剂盒和TNF-α和IL-1β反转录试剂盒购自美国Thermo scientific公司,SYBR qPCR试剂盒购自美国Invitrogen公司。mTOR、p-mTOR、NFκB、p-NFκB、Bcl-2、Mcl-1、β-Actin抗体以及鼠、兔二抗购自美国CellSignal公司。其他试剂以及仪器设备由本实验室提供。
-
分别取对数生长期非小细胞肺癌A549和NCI-H460细胞,用胰酶-EDTA消化并计数,将100 μl的细胞混悬液接种于96孔培养板使每孔的细胞数约为5×103个/100 μl。待24 h后细胞贴壁,加入阿司匹林或/和阿托伐他汀(先用DMSO溶解再用DMEM培养液稀释成不同浓度)。使与细胞作用的阿司匹林浓度分别为0、5、50、100、150、200 μmol/L,阿托伐他汀浓度分别为0、1、2、5、10、20 μmol/L。每个浓度设3个复孔,对照组加入DMEM培养液。置于37 ℃、5% CO2培养箱72 h后,加入10 μl MTS溶液(临用前用PBS溶解,pH7.4,质量浓度为5 g/L),再置培养箱继续培养1 h,于酶标仪490 nm处检测吸光度值(A值)。
细胞增殖存活率(%)=(实验孔A值/对照孔A值)×100。
-
分别将A549和NCI-H460细胞悬液接种于两个6孔培养板中,保证每孔细胞数约为1×105个/孔,次日待细胞贴壁铺满后进行划痕,PBS清洗一遍后更换无血清培养液。根据MTS实验结果选取了浓度为100 μmol/L阿司匹林、5 μmol/L阿托伐他汀以及二者联合作为处理药物,共设3个给药组和1个对照组进行实验,24 h后观察划痕距离变化并在显微镜下拍照,与给药前进行比较。
-
以1×105个/孔接种NCI-H460细胞于6孔板中,同样设置有浓度为100 μmol/L阿司匹林、5 μmol/L阿托伐他汀以及二者联合的3个药物处理组和1个对照组,并给予对应浓度的药物处理细胞,对照组不作处理。继续培养72 h后收集细胞,PBS洗细胞3次,加入细胞裂解液(含20 mmol/L Tris-Hcl/pH 7.5、1% Triton、150 mmol/L NaCl、1 mmol/L EDTA、2.5 mmol/L 无水焦硫酸钠、1 mmol/L β-甘油磷酸盐、1 µg/ml亮肽素和1 mmol/LPMSF),用细胞刮刀将细胞刮下,4 ℃冰浴中超声破碎细胞,待细胞膜破碎反复涡旋后,于4 ℃、12 000 r/min离心,取上清,BCA法蛋白定量后样品经SDS-PAGE分离,蛋白电转至PVDF膜,将PVDF膜在5%牛奶中封闭1 h,加入1∶1 000的一抗,4 ℃孵育过夜,PBS洗涤,加入酶标二抗室温1 h,PVDF膜经ECL化学发光底物检测蛋白。
-
以1×105个/孔接种NCI-H460细胞于6孔板中,设置有浓度为100 μmol/L阿司匹林、5 μmol/L阿托伐他汀以及二者联合的3个药物处理组和1个对照组。次日待细胞贴壁换培养液给予对应浓度的药物处理细胞,对照组不作处理,继续培养72 h后收集细胞。采用RNA提取试剂盒(上海博彩)提取细胞总RNA,按照TNF-α和IL-1β反转录试剂盒说明书(美国Thermo scientific公司)操作进行反转录。得到的cDNA按照SYBR Green PCR Master Mix试剂盒说明书进行实时定量PCR检测。每个PCR反应体系包括:SYBR Green PCR Master Mix 10 μl、cDNA模板2 μl、10 μmol/L上下引物(表1)各0.4 μl以及7.2 μl去离子水,总反应体积为20 μl,经7300 qRT-PCR系统(Applied Biosystems)检测,反应循环参数为95 ℃ 5 s、60 ℃ 35 s共40个循环。基因的表达用ΔΔCt法计算,以β-actin作为内参。
引物名称 引物序列(5’-3’) TNF-α 上游:ACCCTCACACTCAGATCATTCTTCTC 下游:CAGATTGACCTCAGCGCTGAGTTC IL-1β 上游:CCAGCAGGTTATCATCATCATCC 下游:CTCGCAGCAGCACATCAA β-actin 上游:TCACCCACACTGTGCCCATCTACGA 下游:CATCGGAACCGCTCGTTGCCAATAG -
使用SPSS 16.0软件进行数据处理,所有数据以(
$ \bar{x}\pm s $ )表示,采用t检验进行两组间差异比较,P<0.05为显著差异。
1.1. 材料
1.2. MTS法检测细胞增殖活性
1.3. 细胞划痕实验
1.4. Western blotting检测蛋白表达
1.5. 实时定量PCR分析(qRT-PCR)实验
1.6. 统计处理
-
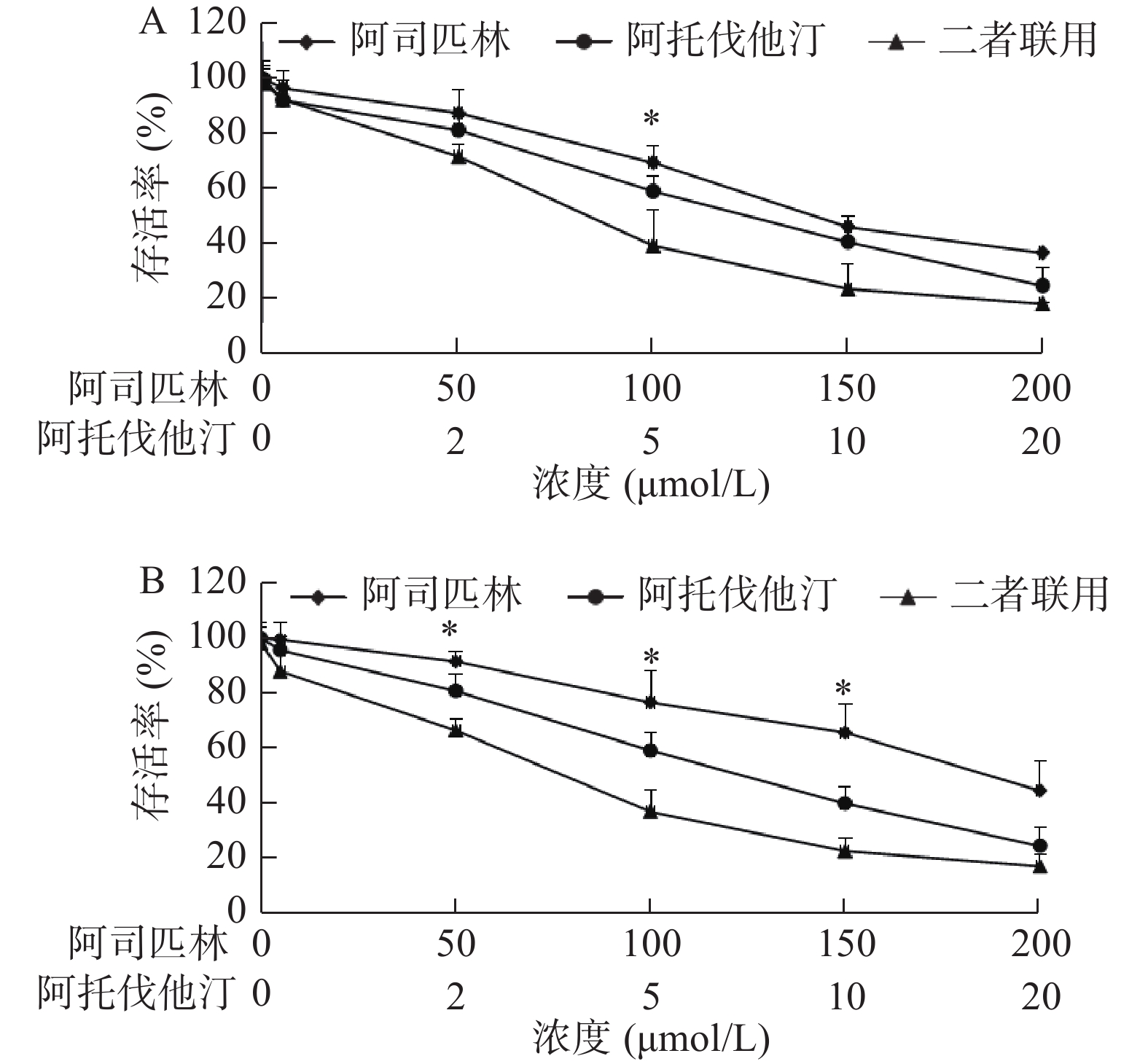
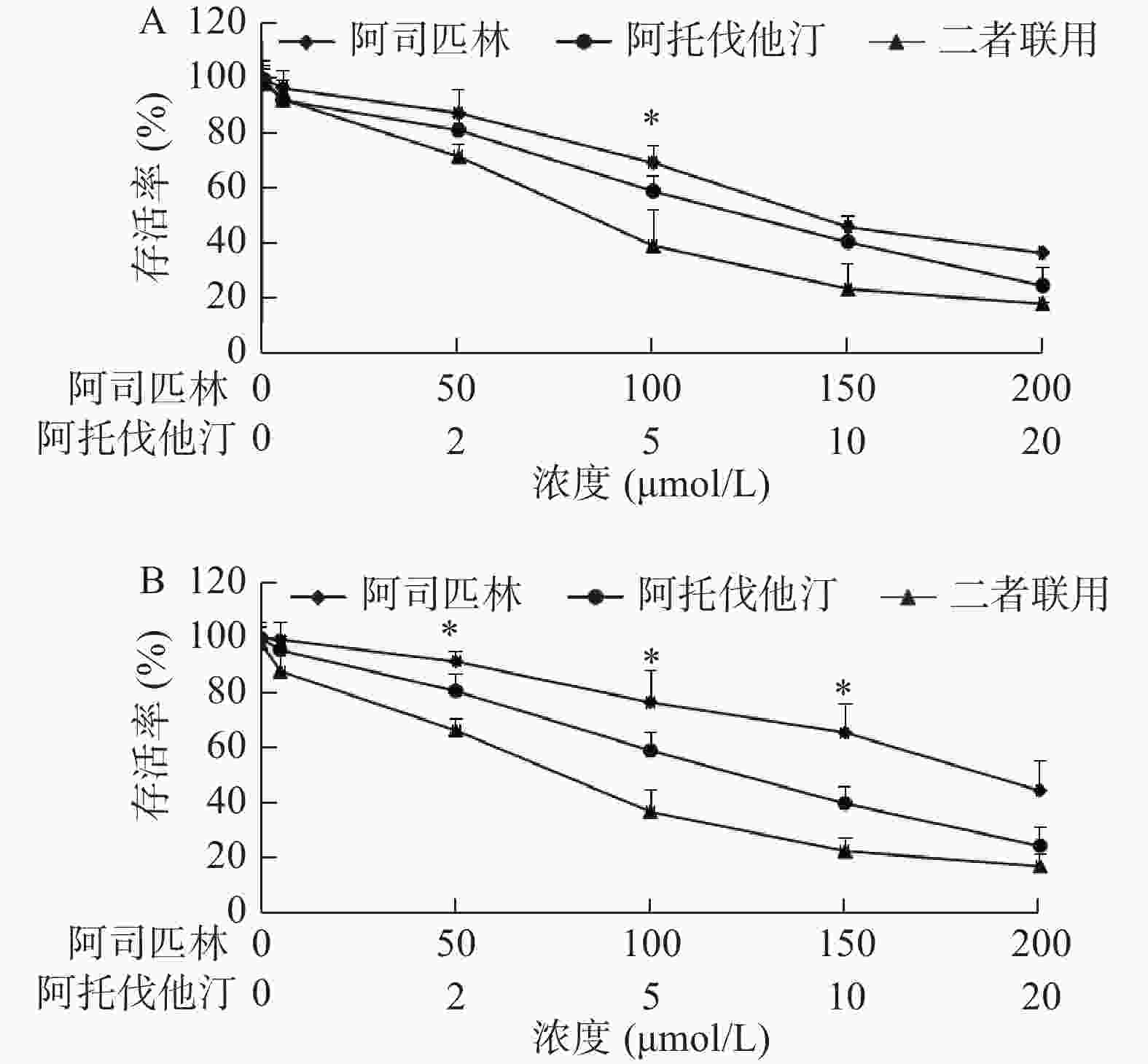
我们选取了浓度分别为0、5、50、100、150、200 μmol/L的阿司匹林和0、1、2、5、10、20 μmol/L的阿托伐他汀处理A549和NCI-H460细胞,72 h后采用MTS法测定两种细胞的增殖活性,以浓度为横坐标,细胞存活率为纵坐标绘制曲线(图1)。结果显示:阿司匹林和阿托伐他汀单用(浓度分别≥100和5 μmol/L)或二者联用均能明显抑制A549和NCI-H460细胞增殖,且二者联用抑制作用大于单用。例如,100 μmol/L阿司匹林和5 μmol/L阿托伐他汀,以及二者联用处理A549细胞,72 h后存活率(与对照孔比较)分别为(69.64±5.95)%、(59.31±5.66)%和(39.51±13.22)%,联合与单用比较,差异有统计学意义(P<0.05)。同样相同浓度的阿司匹林和阿托伐他汀以及二者联用处理NCI-H460细胞,72 h后与存活率(与对照孔比较)分别为(76.73±11.30)%、(59.13±6.61)%和(36.82±8.15)%,且联合与单用比较,差异亦有统计学意义(P<0.05)。
-
根据MTS实验结果,选取了浓度为100 μmol/L阿司匹林、5 μmol/L阿托伐他汀以及二者联合作为处理药物,共设3个给药组和1个对照组进行实验,24 h后观察划痕距离变化并在显微镜下拍照(图2)。结果显示:与给药前比较,阿司匹林、阿托伐他汀以及二者联合均较明显抑制了两种细胞的迁移与侵袭,且二者联合的抑制作用显著增强。
-
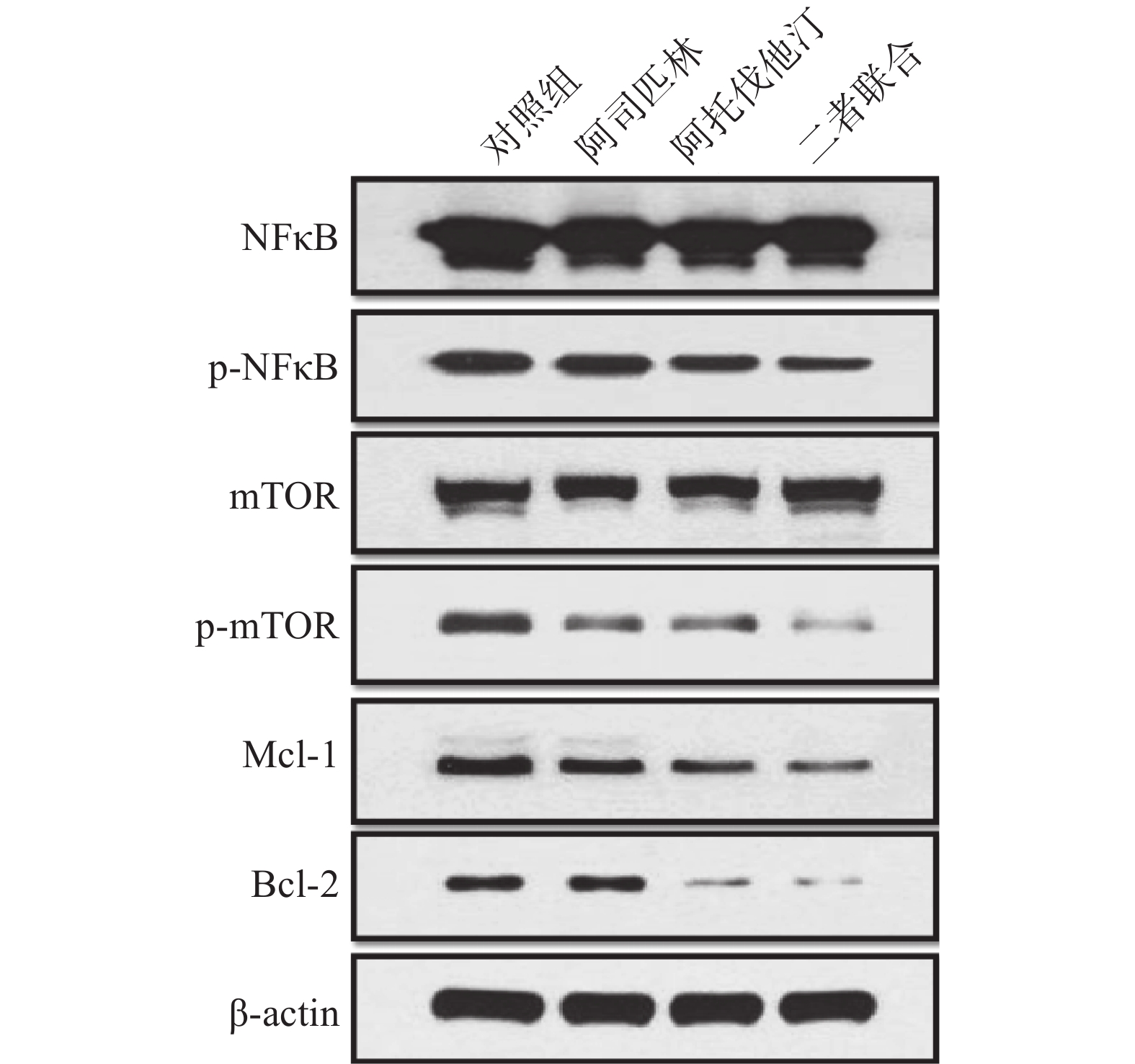
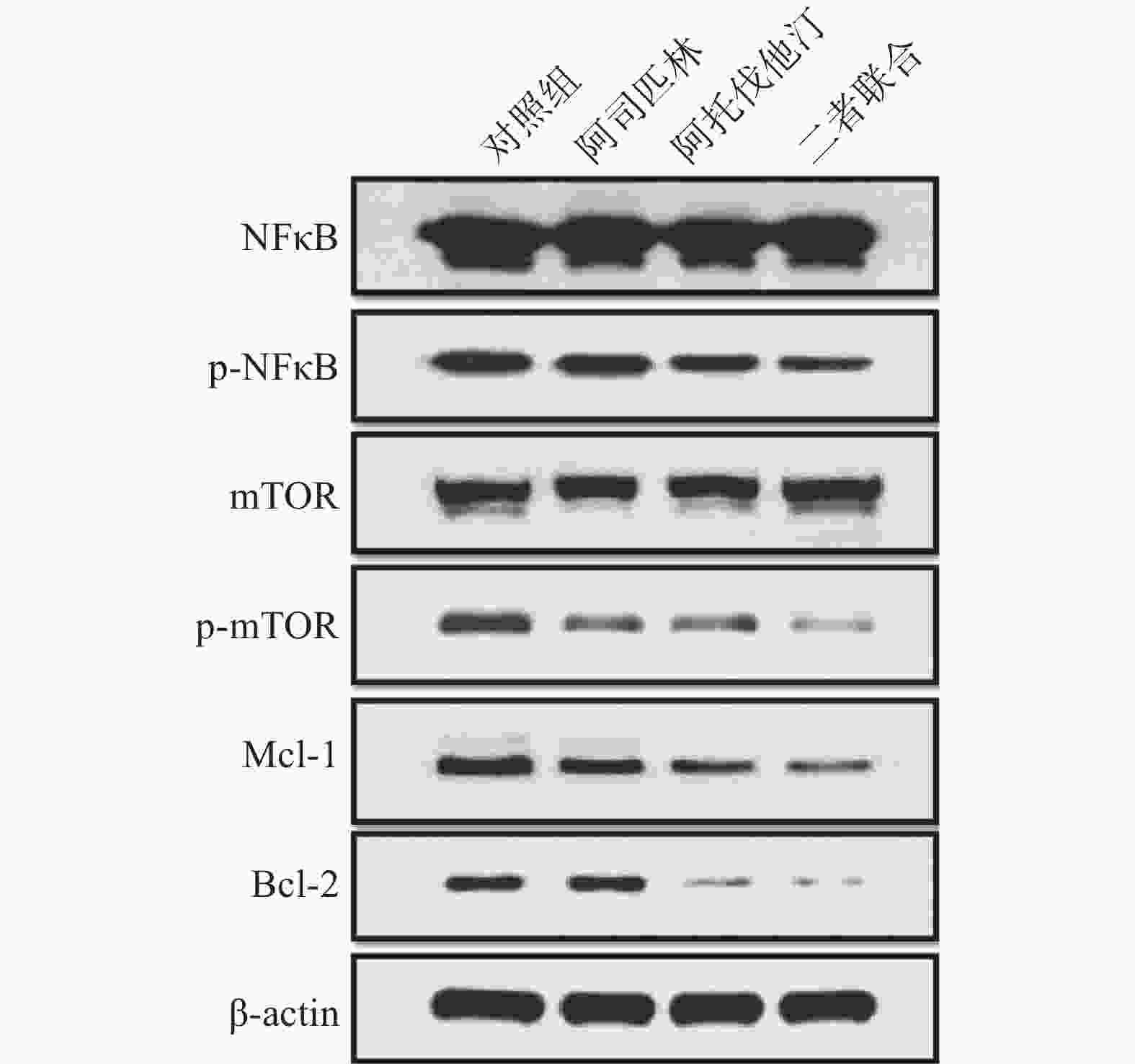
选取浓度为100 μmol/L阿司匹林、5 μmol/L阿托伐他汀以及二者联合作为处理药物,共设3个给药组和1个对照组处理NCI-H460细胞,72 h后收集细胞。细胞裂解提取蛋白并采用BCA法测定总蛋白,采用Western blotting法检测相关蛋白的表达,结果如图3。
结果显示:100 μmol/L阿司匹林、5 μmol/L阿托伐他汀以及二者联合均可以抑制p-NFκB、p-mTOR以及抗凋亡因子Mcl-1和Bcl-2的蛋白表达,且与单药相比,二者联合抑制效应增强,表明阿司匹林和阿托伐他汀联合可以产生协同抑制作用。但与对照组比较,给药组对总NFκB和总mTOR的蛋白表达均无明显影响。
-
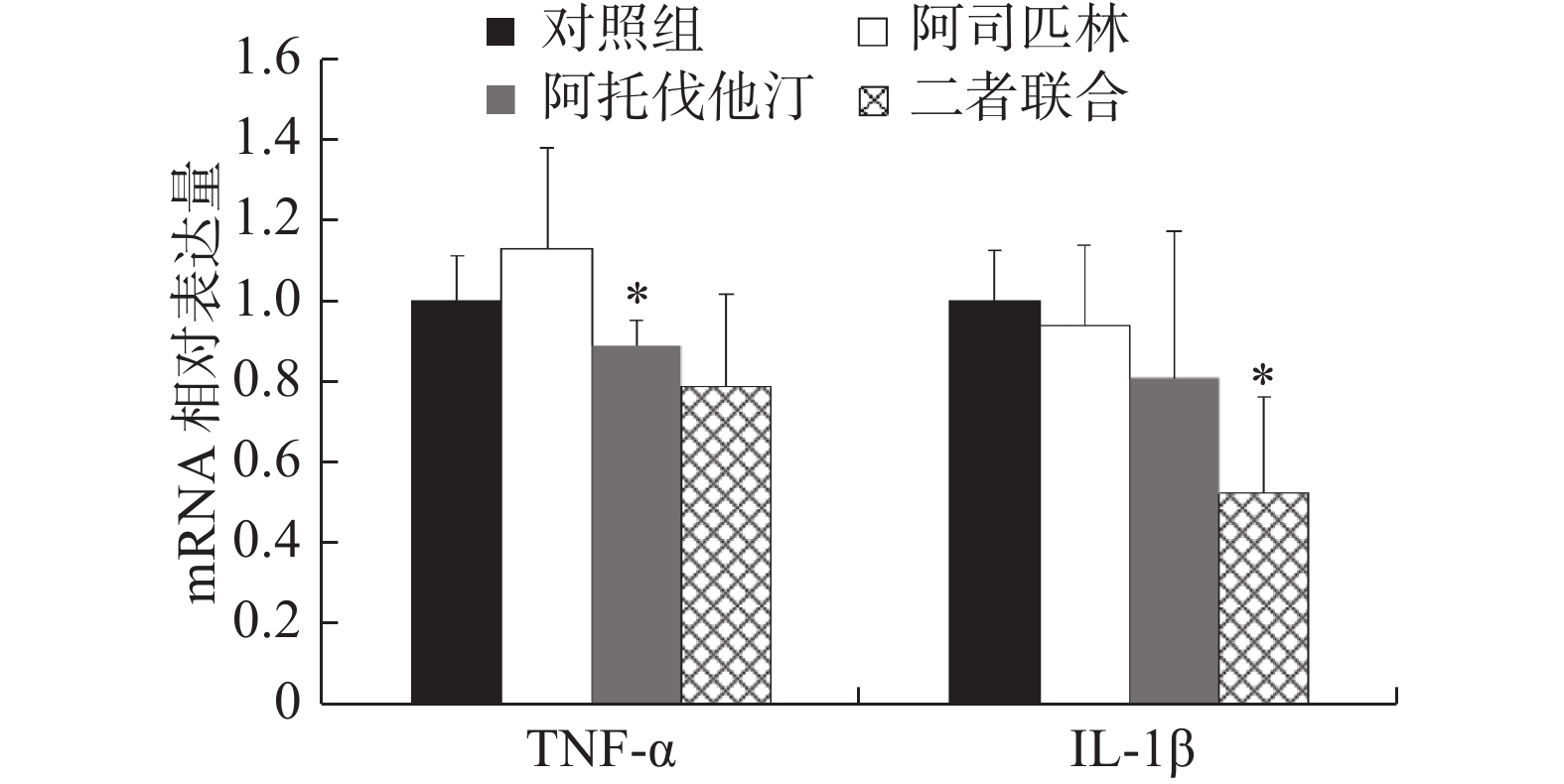
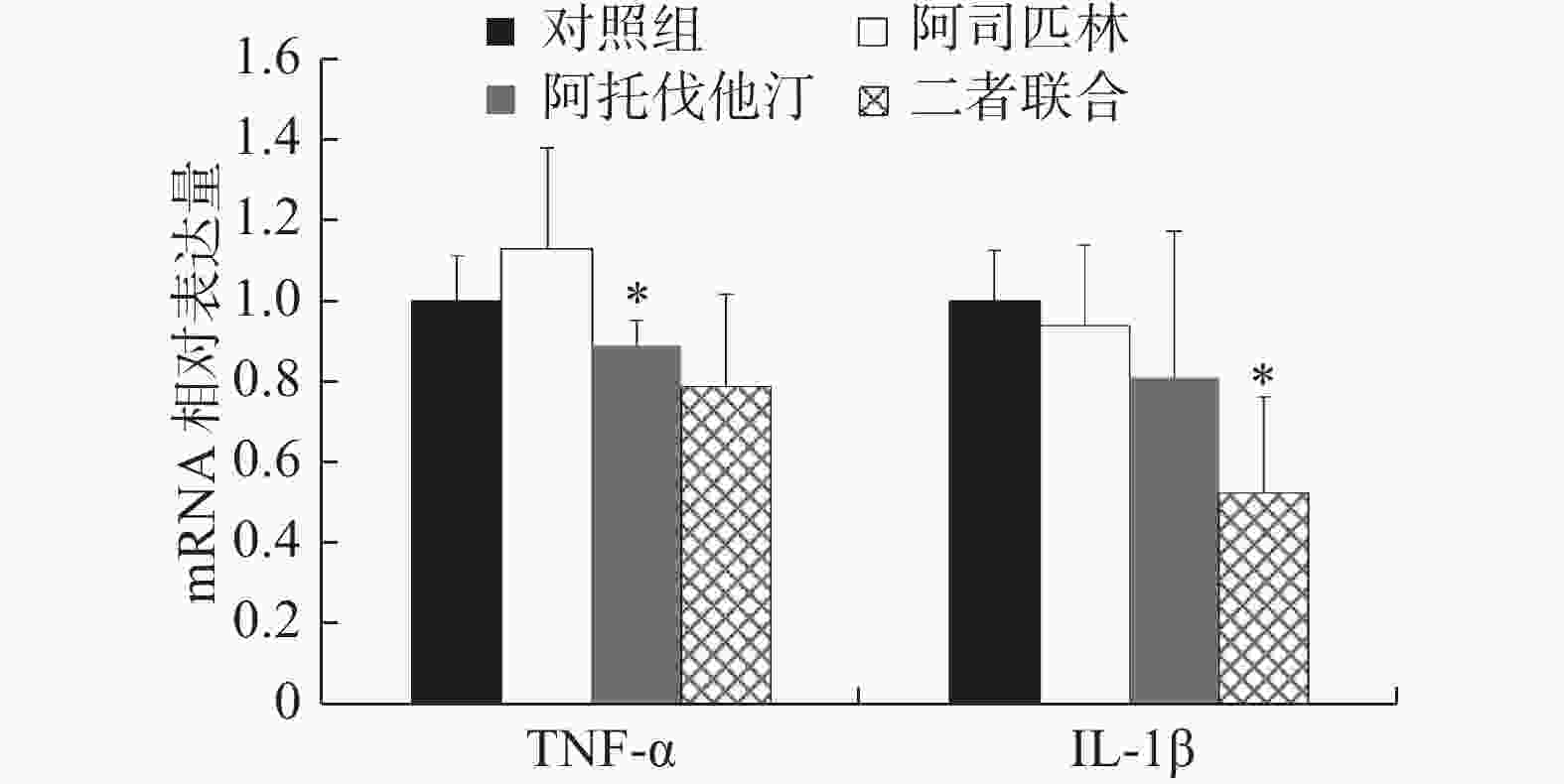
同样,选取浓度为100 μmol/L阿司匹林、5 μmol/L阿托伐他汀以及二者联合作为处理药物,共设3个给药组和1个对照组处理NCI-H460细胞,72 h后收集细胞。提取细胞总RNA,按照试剂盒说明书操作对TNF-α和IL-1β进行实时定量PCR检测,结果如图4。
结果显示:与对照组比较,阿司匹林组、阿托伐他汀组以及二者联合处理组NCI-H460细胞中TNF-α表达量分别是对照组的(1.13±0.25)、(0.89±0.06)和(0.79±0.23)倍,IL-1β表达量分别是对照组的(0.94±0.20)、(0.81±0.36)和(0.52±0.24)倍。其中,阿托伐他汀组TNF-α和联合处理组IL-1β的表达,与对照组比较差异存在统计学意义(P<0.05),尤其是联合处理组IL-1β的表达差异最为明显,与对照组比较下降了近50%。
2.1. 阿司匹林、阿托伐他汀以及二者联合对A549和NCI-H460细胞增殖的影响
2.2. 阿司匹林、阿托伐他汀以及二者联合对A549和NCI-H460细胞迁移与侵袭的影响
2.3. 阿司匹林、阿托伐他汀以及二者联合对NCI-H460细胞信号调控因子相关蛋白表达的影响
2.4. 阿司匹林、阿托伐他汀以及二者联合对NCI-H460细胞TNF-α和IL-1β mRNA表达的影响
-
流行病学数据显示,服用阿司匹林或者阿托伐他汀的人群出现了肺癌患病率降低或者肿瘤发生发展的延缓。譬如,Van Dyke等[6]发现成人加强剂量的阿司匹林可以降低55~64岁妇女罹患NSCLC的概率;Jiang等[7]报道,标准剂量阿司匹林(>325 mg)服用5年以上降低了肺癌事件的发生。Lin等[8]在采用倾向评分法对5 118名65岁以上的Ⅳ期NSCLC患者的临床研究发现,服用了阿托伐他汀平均生存时间提高了3个月,并且得出结论阿托伐他汀提高了Ⅳ期NSCLC患者的存活率。这些报道均提示阿司匹林和阿托伐他汀对肺癌具有直接的抑制效应,然而两药联合使用时是否可以表现出协同抗癌活性,目前报道较少,He等[9]研究显示阿司匹林和阿托伐他汀对前列腺癌细胞的增殖有联合抑制作用,但对肺癌细胞的作用未见报道。据此,我们选取了非小细胞肺癌A549和NCI-H460细胞,在体外研究阿司匹林联合阿托伐他汀对它们的抑制效应及其相关作用机制。
首先,我们采用MTS法检测了阿司匹林和阿托伐他汀单用及联合对A549和NCI-H460细胞增殖的影响,结果显示(图1):当阿司匹林和阿托伐他汀的浓度分别大于100和5 μmol/L时均能明显抑制A549和NCI-H460细胞增殖,且二者联用抑制作用明显大于各自单用。他汀类药物在体内外可以抑制癌细胞(包括NSCLC)的增殖已有文献报道[10-12],该结果与之相符。但阿司匹林在体外抑制NSCLC细胞的增殖,以及联合阿托伐他汀发挥协同抑制作用,文献未见报道。所以,本实验结果提示阿司匹林联合阿托伐他汀可以发挥协同抑制NSCLC细胞增殖效应。其次,我们在细胞划痕实验中观察到100 μmol/L阿司匹林和5 μmol/L阿托伐他汀均能抑制A549和NCI-H460细胞的迁移与侵袭,且二者联合抑制作用显著增强(图2),该结果也进一步说明阿司匹林联合阿托伐他汀发挥协同抑制NSCLC细胞效应。
阿司匹林和他汀类药物在临床上广泛应用于预防和治疗心血管疾病,但近年来由于其抗炎作用、抗肿瘤活性的研究发现,且其分子机制研究已经比较成熟,已经无可争议的成为癌症预防的候选药物。通过文献综述我们发现,阿司匹林和他汀类药物发挥抗肿瘤的分子机制有重叠,即均可以直接或间接作用于mTOR和NFκB信号靶点[4,13],影响它们通路下游一些基因和蛋白的表达,进而发挥抑制癌细胞增殖、诱导凋亡、抗血管生成以及转移等一系列抗肿瘤活性。本研究我们在NCI-H460细胞中验证了阿司匹林和阿托伐他汀靶向作用于mTOR和NFκB信号靶点,抑制它们的磷酸化过程(即抑制活化),且能够抑制它们信号转导下游抗凋亡基因Mcl-1和Bcl-2的蛋白表达。同时还发现当阿司匹林和阿托伐他汀联合应用时,对这些信号通路中这些蛋白的表达下调作用更加明显,提示二者联合发挥了协同作用(图3)。另外,文献报道他汀类药物可以通过作用炎症调控因子NFκB影响细胞内一些重要炎症因子(如TNF-α、IL-8等)的生成[14-15],同样,我们在研究中也发现阿托伐他汀降低了NCI-H460细胞中炎症因子TNF-α的mRNA表达,对IL-1β的mRNA表达也有抑制作用但差异没有统计学意义(与对照组比较P>0.05,图4),阿司匹林未发现有相同的作用,但二者联合显著降低了IL-1β的mRNA水平,与对照组比较下降近50%。此结果提示阿司匹林和阿托伐他汀联合应用可能对某些促炎因子(如IL-1β)的影响有协同作用。
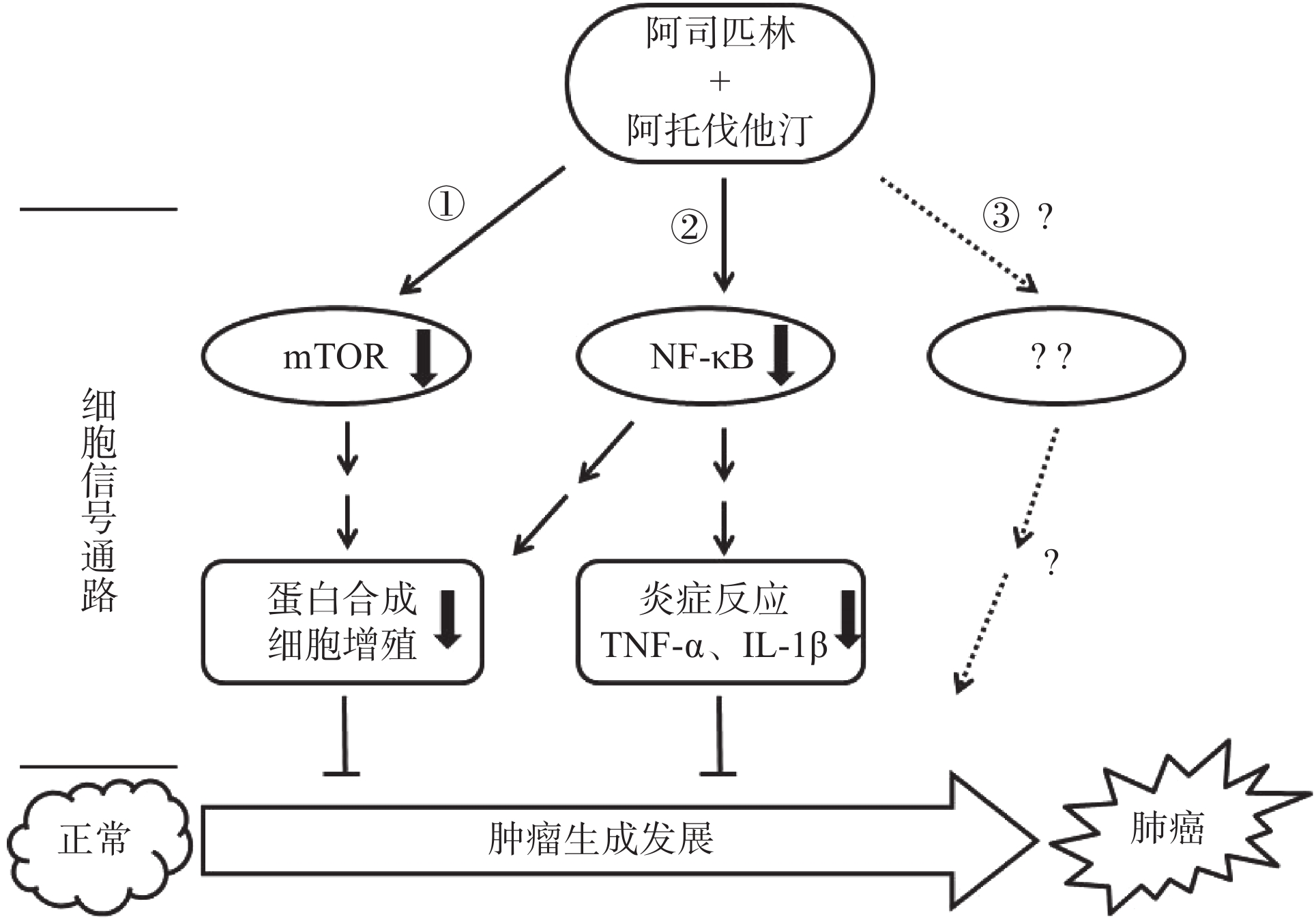
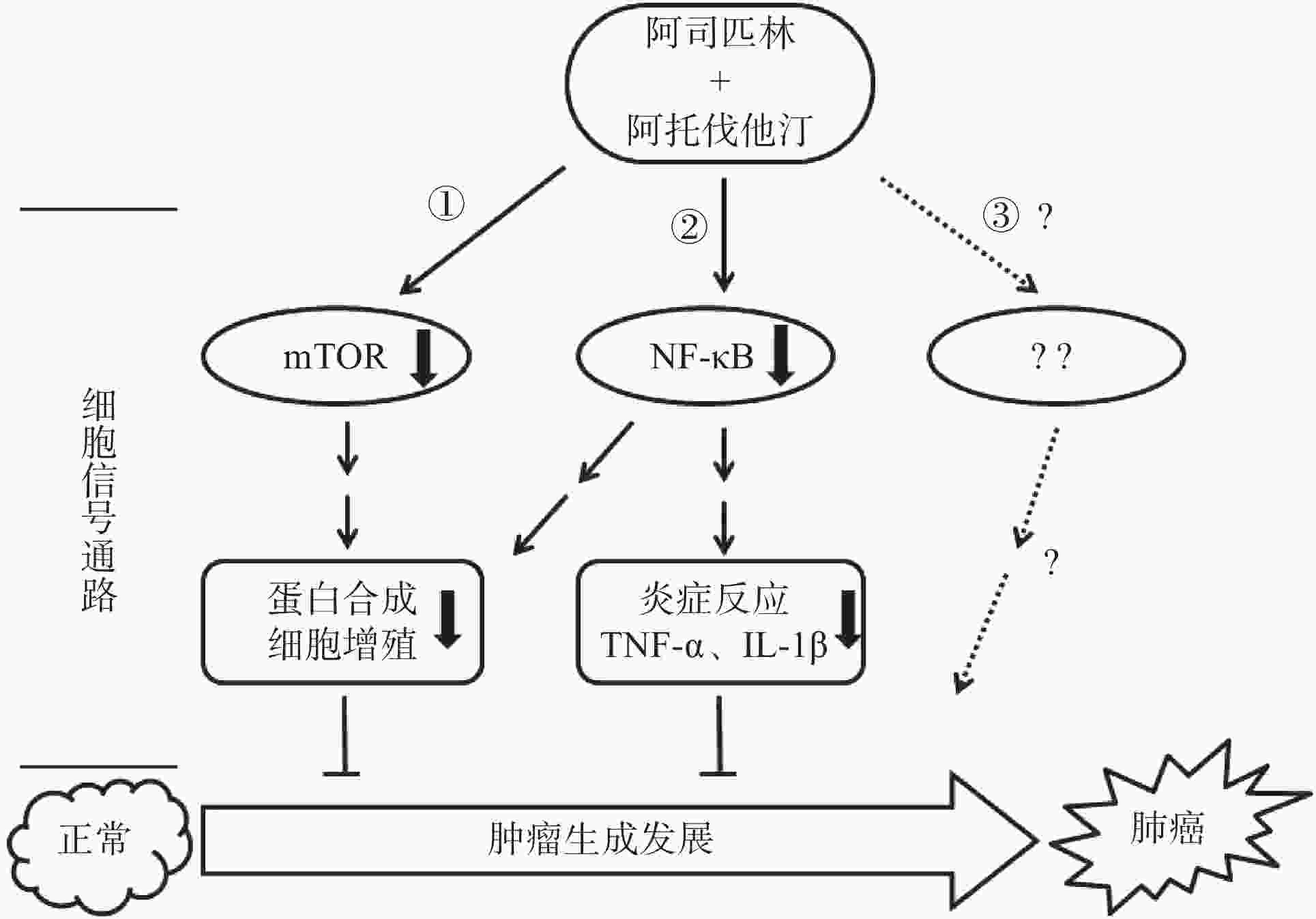
综上所述,阿司匹林联合阿托伐他汀可以协同抑制非小细胞肺癌A549和NCI-H460细胞的增殖,其机制为共同作用于mTOR和NFκB信号靶点并且影响该靶点信号转导下游调控基因的表达,如抗凋亡基因Mcl-1、Bcl-2以及炎症因子TNF-α和IL-1β(图5),但这种协同作用是如何通过作用mTOR和NFκB并调控其下游目的基因来实现的,以及是否还通过作用于新的信号靶点或者其他信号通路(图5路线③)发挥协同抑制作用有待于进一步研究求证。









 DownLoad:
DownLoad: