-
相对于传统纳米载体而言,由红细胞、血小板、干细胞、巨噬细胞、肿瘤细胞以及细菌等构成的新型内源性载体具有生物相容性好、靶向性高的显著优势[1]。但是,该类细胞载体由于自身理化性质导致其无法在特定组织内渗透和摄取。近年来,随着纳米技术与生物技术的交叉融合,研究者从仿生角度出发,通过物理挤出、共孵育和微流体等技术工艺将各类型天然细胞膜与纳米载体内核相结合,制备出体内循环时间长、生物相容性好、胞内细胞器靶向性能高的新型仿生纳米药物载体[2]。本文综述了细胞膜修饰技术的基本原理及几种最为常用的细胞膜修饰纳米载体的特点及应用。
-
细胞膜是由磷脂、糖蛋白、糖脂和蛋白质构成的富有弹性的半透性膜,其中,磷脂双分子层是构成细胞膜的基本结构,其在广泛的生物功能中发挥着重要作用。细胞膜的提取包括膜溶解和膜纯化[3],具体过程由所提取的细胞类型决定。对于无核细胞(如哺乳动物成熟红细胞和血小板),可通过低渗处理或者反复冻融破坏其细胞膜,差速离心去除可溶性蛋白质,然后通过挤压形成由细胞膜构成的囊泡[4]。对于白细胞、癌细胞和干细胞等真核细胞,膜提取过程则相对复杂。首先,需要从组织或血液中分离目标细胞,之后先进行细胞培养[5],再将培养好的细胞通过低渗裂解、机械膜破坏和蔗糖梯度离心相结合的方法去除细胞核和细胞质,从而分离出细胞膜[6]。分离出的细胞膜用等离子缓冲剂彻底清洗后,通过多孔聚碳酸酯膜挤压形成纳米囊泡[7]。
-
内核纳米材料在细胞膜仿生纳米载体技术中至关重要[8]。近年来,各种类型的纳米材料(如聚合物纳米粒、脂质体、二氧化硅、介孔二氧化硅、金粒子、氧化铁、金属-有机骨架、纳米凝胶和黑磷等)已成为细胞膜仿生技术的核心载体。由脂质和聚合物为材料制备的纳米载体因其良好的生物相容性和高载药量,广泛用于制备纳米粒子[9]。无机材料具有成本低、易于进行化学结构修饰及特定光电磁性质的显著优势,可以更好地控制无机颗粒的表面组成、形状和尺寸,并且可以用适当的膜囊进行伪装[8]。最新研究发现,纳米凝胶由于其大孔结构和高负载能力而成为理想的内核载体材料[10];同时,Chen等[11]利用黑磷优良的光热性能、负电位及生物降解性将其与红细胞膜融合制备成为光热治疗剂,结果表明该方法能够有效抑制乳腺癌细胞的生长。
-
目前,常用的生物细胞膜与纳米载体融合方法有薄膜挤压法、超声融合法和电穿孔法。薄膜挤压法借助挤压器通过纳米级聚碳酸酯多孔膜反复挤压,利用机械力使纳米材料与细胞膜相融合[12],该方法工艺简便,但难以大规模制备。超声融合法是将纳米粒子与细胞膜共孵育后利用超声促进细胞膜的包覆,但是该方法生成的载体颗粒均一性较差[13]。微流控电穿孔法将载体各组分在Y形通道中完全混合,通过电穿孔制备出性质稳定、包覆率高的纳米载体[14]。
-
细胞膜纳米粒子的评估包括物理、化学和生物特性3个方面,以确定细胞膜是否成功地包覆在纳米粒子表面。细胞膜包覆的效率取决于纳米粒子的大小、表面电荷和蛋白质组成。细胞膜改变了纳米粒子大小和Zeta电位,透射电子显微镜(TEM)可用于确认细胞膜纳米粒子的形态,与未包覆的纳米粒子相比,粒子直径明显增加。Zeta电位表明了细胞膜包覆前后颗粒表面的电位特征。Fan等[15]以红细胞膜包覆后,颗粒的Zeta电位增加了10mV。另外,可通过动态光散射(DLS)验证细胞膜纳米粒子的粒径分布[16]。细胞膜的仿生功能主要取决于其生物学特性。因此,验证膜的生物活性是至关重要的,从而确保最佳的纳米粒子包覆效能。蛋白免疫印迹法常被用于鉴别包覆于纳米粒上的细胞膜蛋白,从而验证纳米载体的生物特性,Wang等[17]通过该方法证实了红细胞膜特征蛋白CD47存在于被红细胞膜包覆的纳米粒子表面。
-
经天然细胞膜包覆后的纳米粒子生物相容性增强,具有特异性识别靶部位、靶组织和靶细胞的特性。近年来,各种类型的天然细胞已被广泛应用于纳米载体制备,如红细胞、血小板、免疫细胞、癌细胞,甚至细菌膜,以大肠杆菌为膜源,组装构建出具有多种功能的生物复合传递系统,发挥其特殊的靶向和治疗作用。
-
红细胞由于自身生物特性(例如红细胞表面跨膜蛋白CD47),能够在体内不受免疫系统的攻击[18],利用红细胞膜包覆纳米粒子制备成的药物载体具有循环时间长和免疫逃避特性等显著优势[19]。Fang等[20]通过挤压法和超声法将红细胞囊泡与叶酸和核苷靶向适体AS1411相融合,结果显示该方法获得的红细胞膜纳米载体能够显著提高肿瘤靶向性和治疗效果。红细胞膜表面有各种成孔毒素相关特异性受体成分,且具有磷脂双分子层结构,因此有广谱的成孔毒素中和作用且生物相容性好。经红细胞膜包覆的纳米粒子可以吸附多种成孔毒素(如α-溶血素、链状溶血素-O等),并保护红细胞免受溶血[21]。研究显示这些纳米粒子并没有将毒素转移到宿主细胞,确保了生物安全性。同时,将灭活毒素整合到纳米粒子的细胞膜中作为类毒素疫苗,对受到耐甲氧西林金黄色葡萄球菌攻击的小鼠具有明显保护作用,能显著减少相关肺损伤,并抑制脾脏炎症转录因子的激活。通过利用红细胞血液循环时间长和“自我标记”蛋白的特性,这些仿生纳米粒子可以避免免疫细胞对其捕获。
-
血小板具有广泛的抗原和功能蛋白,并在肿瘤转移中发挥重要作用,其被开发成为细胞膜材料[22],可用于多发性骨髓瘤和血栓治疗、动脉粥样硬化检测、癌症治疗以及控制细菌感染。Wei等[23]用血小板膜包覆纳米粒子以清除体循环中的病理性抗体,这一策略被用来治疗免疫性血小板减少性紫癜,能有效地减少血小板的破坏,并保持正常的止血功能。Ávila等[24]研究发现,用红细胞膜和血小板膜的混合物包覆纳米粒子使其成为优良的解毒剂,赋予纳米粒子许多新的生物学特性,包括结合黏附于血小板的病原体(如金黄色葡萄球菌细菌)以及中和成孔毒素(如志贺毒素和金黄色葡萄球菌)。
-
各类免疫细胞(例如巨噬细胞、中性粒细胞、树突状细胞、干细胞及T细胞)能够在机体产生免疫反应的第一时间被招募至特定位置发挥免疫功效[25-26],使其成为包覆纳米载体的理想载体膜。Xuan等[27]研究发现,经巨噬细胞膜包覆的二氧化硅纳米粒子在体内的循环时间明显延长,并能够提高阿霉素的传递效率和抗癌效果。同时,Xuan等[28]用巨噬细胞膜包覆近红外成像探针(Cy7)载金纳米粒子,能够增加药物在肿瘤组织的积聚,在近红外激光照射下,由巨噬细胞膜包覆的金纳米粒子产生的局部热量可高效抑制肿瘤生长,并选择性地消融热辐射区域内的癌细胞。Zhang等[29]用巨噬细胞膜包覆对酸碱度敏感的聚合物纳米粒子,用于肿瘤靶向化疗,具有响应肿瘤微环境刺激的控释特性。其将紫杉醇传递至靶向位置,发现该载体能够使药物在肿瘤低pH值环境中充分释放。中性粒细胞在免疫反应中扮演重要角色[30]。Gao等[31]研究发现,基于中性粒细胞膜纳米囊泡药物递送系统可选择性地靶向于炎症血管部位释药,以逆转急性肺部炎症。Zhang等[32]将人的外周血中性粒细胞膜包覆在聚乳酸-羟基乙酸(PLGA)聚合物芯上,构建炎症治疗平台,从而抑制滑膜炎症并减轻炎症性关节炎受试者的关节损伤。此外,Wei等[33]将人T淋巴瘤细胞(SUP-T1)的细胞膜包覆在聚乳酸-羟基乙酸共聚物(PLGA)上,T细胞膜包覆纳米粒子后可选择性地与HIV的糖蛋白gp120结合,并抑制gp120诱导的对CD4+T细胞的杀伤。当加入到HIV病毒中时,T细胞膜包覆纳米粒子以剂量依赖性的方式有效地中和外周血单个核细胞和人类单核细胞源性巨噬细胞的病毒感染。通过利用天然T细胞功能,T细胞膜包覆纳米粒子作为一种新的抗HIV感染治疗剂显示出巨大的潜力。干细胞作为一种“万能细胞”,在不同发育阶段都具有靶向肿瘤细胞的特性[34]。Cao等[35]研究了一种新型抗癌药物载体,即包封载药空心二氧化硅纳米颗粒的间充质干细胞(MSCs),由于MSCs天然的高肿瘤亲和力,用于携带光敏剂药物并将其输送至乳腺肿瘤,并通过光动力学疗法抑制肿瘤生长。这种新策略解决了光动力疗法中光敏剂药物在肿瘤中积聚的问题,并成功用于肿瘤靶向治疗。
-
癌细胞具有无限复制潜能、免疫逃逸、循环时间长和同源结合能力等独特功能[36],从而使癌细胞膜成为设计抗癌纳米递药系统的包覆材料[37]。Chen等[38]构建了一个核心−外壳的纳米结构ICNP,由吲哚青绿(ICG)和人乳腺癌细胞膜(MCF-7)外壳组成。ICNP表现出对癌细胞的特异性同源靶向性,具有良好的单分散性、较好的光热响应和优异的荧光/光声成像特性。得益于来自癌细胞膜的同源结合黏附分子的功能化,ICNP显著促进细胞内吞和体内同源靶向肿瘤积聚; 在近红外激光照射下,ICNP表现出高效的光热疗法来根除异种移植肿瘤,具有癌细胞膜同源性的ICNP可以作为肿瘤靶向成像和光疗的仿生纳米平台。Yang等[6]将甘露糖修饰的黑色素瘤细胞膜包覆在一种负载受体7激动剂(咪喹莫特)的PLGA纳米粒子上组成的抗癌疫苗,可使其被树突状细胞更好地摄取,从而触发机体对癌细胞的特异性免疫反应,延缓了肿瘤的发展恶化。Sun等[39]用4T1乳腺癌细胞膜包覆负载紫杉醇的聚合物纳米粒子,结果发现该载体可以选择性地积聚在原发性肿瘤和转移性结节中,血液清除速度较慢,并且显著抑制转移性肿瘤和原发性肿瘤的生长。其研究还可能通过使用癌症患者的肿瘤细胞膜作为药物载体,为转移癌的个性化治疗开辟一条新途径。以上研究可以发现,将不同的肿瘤细胞膜开发为膜包覆材料,利用其特异性的靶向能力,在肿瘤治疗方面具有广阔的应用前景。
-
与基于细胞的仿生系统一样,细菌的蛋白质外壳与功能性纳米颗粒结合,广泛应用于药物输送、肿瘤成像和治疗[40]。鉴于其高水平的免疫原性蛋白和佐剂特点,细菌细胞膜在病原体相关分子靶点介导和适应性免疫系统激活的环境中可用于纳米粒子修饰[41]。Gao等[42]用大肠杆菌衍生膜包覆30nm的金纳米粒子并将其注射于小鼠皮下,结果发现这些粒子能被运送到近端引流淋巴结,并能激活树突状细胞。同时,接种了大肠杆菌膜纳米粒子的小鼠比仅接种了外膜囊泡的小鼠表现出更高的抗体亲和力和IFN-γ及IL-17水平,该结果证实被大肠杆菌包覆的纳米粒子载体能够特异性激活T细胞免疫反应。Zhang等[43]将光敏剂原卟啉IX和抗菌肽2结合起来,制备出对细菌光动力疗法失活的PPK颗粒,这些颗粒能够通过静电相互作用和膜渗透结合在细菌细胞上,进而破坏细菌细胞膜组织起到杀菌作用。
细菌细胞膜包覆纳米粒子不仅能简化细菌载药系统,而且能构建纳米级别的靶向载药系统。但是,该方法也对生物安全性提出一些需研究问题,如细菌细胞膜的抗原特性,是否会引起感染相关问题仍需进一步的研究。
-
细胞膜仿生纳米粒子将包覆材料和内源性生物材料进行融合,进一步激发从生物学角度设计新型给药系统的创新发展。细胞膜包覆的纳米粒子将在体内药物递送、生物成像和疾病治疗,特别是癌症的诊断和治疗方面显示出许多优势。然而,使用单一类型或常规类型的细胞膜作为包覆材料可能会限制其功能多样性,混合类型的细胞膜与纳米载体共同构建高选择性的靶向药物输送系统具有广泛的研究价值。结合全面的生物特性和功能,整合纳米材料将增强治疗性纳米粒子的体内性能。例如,可以考虑将由抗体、脱氧核糖核酸/核糖核酸、肽、蛋白质和酶组成的简易配体修饰于细胞膜,从而增强精准靶向性能,甚至是协同的治疗作用。尽管细胞膜仿生技术尚未完全实现临床应用,但其明显的优势和丰富的细胞膜来源为其工业化生产和在个体化精准医学中的实施提供了坚实的基础。
Progress in preparation and application of biomimetic nano carriers for cell membrane
-
摘要: 由有机或无机纳米材料制备的药物载体系统广泛用于药物靶向递送和疾病的诊断治疗研究。但其存在靶向性差、体内循环时间短、生物相容性欠佳亟需提高等问题。仿生纳米药物系统是以不同种类的细胞膜修饰纳米载体,利用内源性的细胞膜提高载体的体内生物相容性、实现更精准的靶向、甚至由细胞自身的免疫原性产生免疫治疗作用。对细胞膜仿生纳米载体技术的原理、方法及其靶向机制和治疗作用作一综述,为新型给药系统研究提供思路。Abstract: Nanocarriers prepared from organic or inorganic materials are widely used in drug targeting system and diagnosis and treatment of disease. However, there are some problems, such as poor targeting, short circulation time in vivo and improvement in the biocompatibility. Biomimetic nanocarriers has carried out research on the issues, which based on different kinds of cell membrane for the nanocarriers modification, endogenous biofilm improving the biocompatibility of carriers in vivo, more accurate targeting, and even producing immunotherapeutic effect. The principle, method, targeting mechanism and therapeutic effect of biomimetic nano carrier technology of cell membrane have been reviewed in this paper, which provide a new direction for the research of new drug delivery system.
-
Key words:
- cell membrane /
- biomimetic nanocarriers /
- drug delivery system /
- biomaterials
-
放线菌以其能产生结构新颖且有良好生物活性的先导化合物而备受关注[1],一直被认为是天然药物的重要生产者,其主要结构类型包括聚酮、生物碱、多肽和萜烯类化合物等,同时涵盖了多种多样的生物活性如抗菌、抗寄生虫、免疫调节、抗炎、抗癌等[2-4],这突显了放线菌具有不可预估的药物开发潜力。
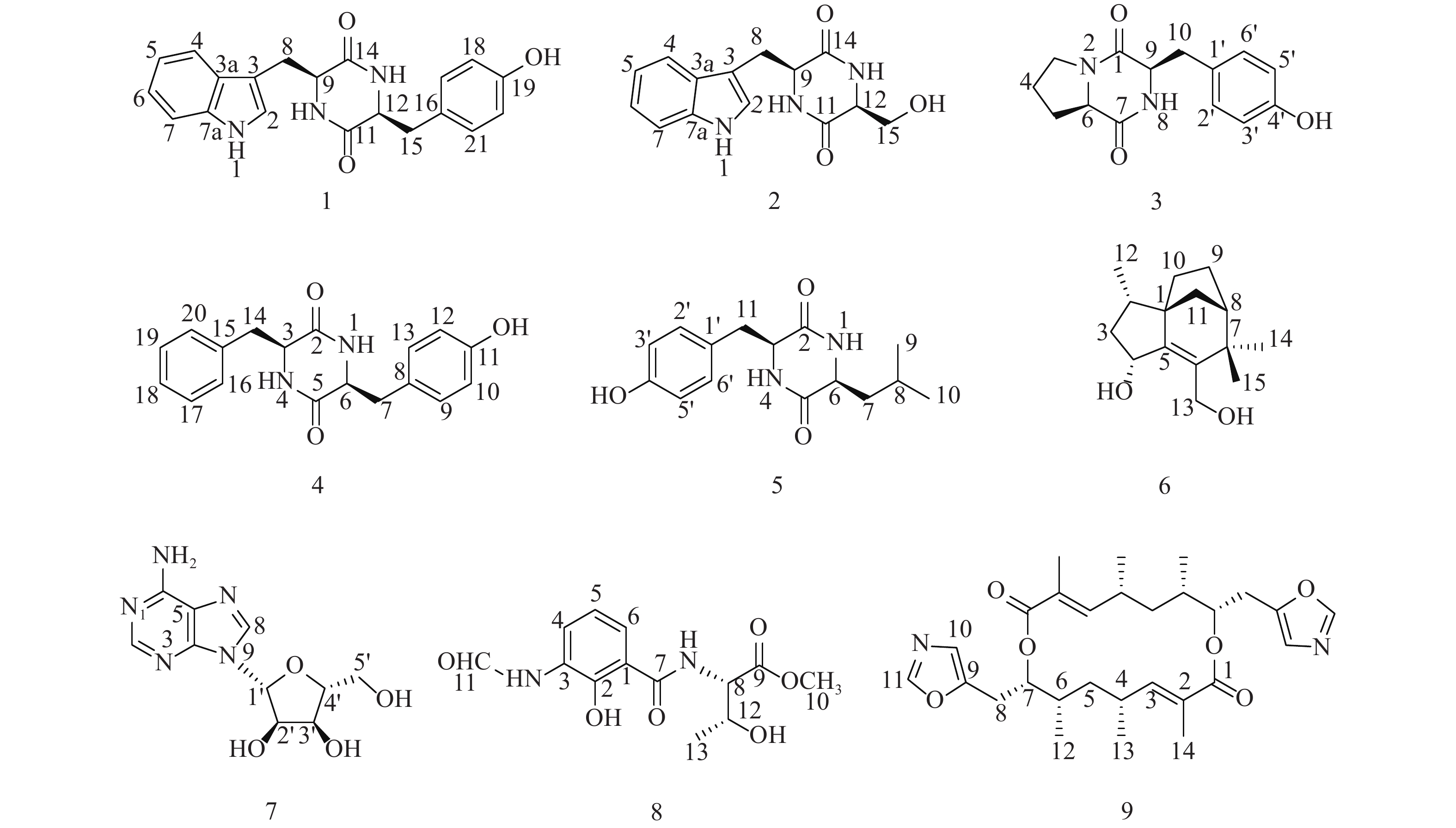
随着研究的深入,陆地和普通环境中的资源日趋枯竭,很多微生物及其次生代谢产物被重复开发和提取分离,发现新活性分子的几率愈来愈低,开发创新药物的难度越来越大[5-6];而极地极端的生态环境造就的微生物具有产生更为特别的化学骨架和活性次生代谢产物的能力,是新型药源分子的重要来源。本文以采自北极楚克奇海域海绵共附生放线菌Streptomyces sp. LHW11-07为研究对象,从其发酵浸膏中分离鉴定了9个单体化合物1~9(图1),其中化合物1和2为该属内首次分离得到。
1. 材料和方法
1.1 实验仪器与试剂
AMX-600型核磁共振仪(德国Bruker公司);Xevo G2-XS Q-TOF液质联用仪、1525/2996, 2998型高效液相色谱仪(美国Waters公司);半制备型HPLC色谱柱(Atlantis Prep T3,美国Waters公司;YMC C18,日本YMC公司);中压柱色谱仪(法国Interchim公司);恒温振荡培养箱(上海一恒科学仪器有限公司);N-1000型旋转蒸发仪(上海爱郎仪器有限公司);反相ODS硅胶和Sephadex LH-20柱色谱填料(Pharmacia公司);正相硅胶(200-300目)和TLC薄层板(烟台江友硅胶开发有限公司);分析级试剂(上海化学试剂公司);色谱级试剂(德国Merck公司);氘代试剂(美国剑桥同位素实验室公司)。
1.2 菌株的来源及鉴定
菌株分离于北极海域来源的海绵样本,经16S rRNA基因序列鉴定为Streptomyces sp.,编号为LHW11-07,菌种保存于上海交通大学医学院附属仁济医院药学部海洋药物研究中心。
1.3 菌株的大发酵
培养基为ISP2:葡萄糖(4 g/L)、酵母提取物(4 g/L)、麦芽糖提取物(10 g/L)以及海盐(25 g/L),加水溶解后调节pH为7.2~7.4,分装后高压灭菌20 min (121 ℃),冷却备用。
挑取Streptomyces sp. LHW11-07单菌落至1级种子培养基里(100 ml ISP2培养基至250 ml三角瓶),置于30 ℃,220 r/min的恒温摇床培养3 d,得1级种子液;将1级种子液按5%接种量接到2级种子培养基里(150 ml ISP2培养基至500 ml三角瓶),置于30 ℃,220 r/min的恒温摇床培养3 d,得2级种子液;将2级种子液按5%接种量接到大发酵培养基里(700 ml ISP2培养基至2 L三角瓶),置于30 ℃,220 r/min的恒温摇床培养7 d,共得到发酵液50 L。
1.4 发酵产物的提取与分离
菌株培养7 d后,用等体积的乙酸乙酯萃取3次,合并乙酸乙酯提取液,减压浓缩得粗浸膏9.8 g。粗浸膏先经凝胶柱分离,二氯甲烷:甲醇(1∶1)的混合溶剂进行洗脱,得到组份Fr.1~Fr.7。
组份Fr.4经正相中压柱色谱分离(二氯甲烷:甲醇100:0~0:100),得到组份Fr.4a~Fr.4j。组份Fr.4d再经反相中压柱色谱分离(10%~100%乙腈水),得到组份Fr.4d1~Fr.4d8,Fr.4d2和Fr.4d3用反相半制备HPLC纯化(35%甲醇水,YMC C18),分别得到化合物1 (12 mg, tR=26 min)和2 (47.6 mg, tR=30 min);Fr.4d6用反相半制备HPLC纯化(49%甲醇水,YMC C18),得到化合物3 (8.4 mg, tR=23 min)和4 (2.0 mg, tR=30 min);组份Fr.4g再经凝胶柱纯化,洗脱剂为正己烷:二氯甲烷:甲醇(4∶5∶1),得到组份Fr.4g1~Fr.4g11,其中Fr.4g3用反相半制备HPLC纯化(25%乙腈水,YMC C18),得到化合物5 (1.2 mg, tR=18 min)。
组份Fr.7经反相中压柱色谱分离(10%~100%乙腈水),得到组份Fr.7A~Fr.7D。组份Fr.7A用反相半制备HPLC纯化(20%乙腈水,YMC C18),得到化合物6 (18 mg, tR=19 min)和7 (3.4 mg, tR=25 min);组份Fr.7B经正相中压柱分离(石油醚:丙酮100:0~0:100),得到组份Fr.7B1~Fr.7B9,Fr.7B4用反相半制备HPLC纯化(88%乙腈水,Atlantis Prep T3),得到化合物8 (12 mg, tR=23 min)和9 (8.4 mg, tR=26 min)。
2. 结构鉴定
化合物1:淡黄色固体,ESI-MS显示准分子离子峰m/z 372 [M+Na]+。1H-NMR (600 MHz, DMSO)显示在低场区有吲哚环的特征信号δH 7.00 (1H, s, H-2),7.49 (1H, d, J=8.0 Hz, H-4),7.02 (1H, t, J=7.6 Hz, H-5),7.05 (1H, t, J=7.6 Hz, H-6),7.32 (1H, d, J=8.0 Hz, H-7);4个活泼氢质子信号δH 10.89 (1H, d, J=2.6 Hz, NH-1),7.83 (1H, d, J=3.0 Hz, NH-10),7.62 (1H, d, J=3.0 Hz, NH-13),9.20 (1H, s, OH-19);4个芳香质子信号δH 6.53 (2H, d, J=8.4 Hz, H-17, H-21),6.59 (2H, d, J=8.4 Hz, H-18, H-20),提示分子中有1个对位二取代的苯环;在高场区有两组亚甲基质子信号δH 2.80 (1H, dd, J=14.5, 4.5 Hz, H-8),2.43 (1H, ov, H-8),δH 1.83 (1H, dd, J=13.4, 6.9 Hz, H-15),2.47 (1H, ov, H-15),两个次甲基质子信号δH 4.01 (1H, m, H-9),3.95 (1H, m, H-12)。13C-NMR (150 MHz, DMSO)结合DEPT谱表明其有20个碳信号,2个酮羰基碳δC 166.7,166.2,14个芳香碳,2个亚甲基碳δC 30.0,40.0和2个次甲基碳δC 55.9,55.2。对其碳信号进行归属:δC 118.7 (C-2)、108.9 (C-3)、127.5 (C-3a)、118.4 (C-4)、120.8 (C-5)、124.3 (C-6)、111.3 (C-7)、136.0 (C-7a)、30.0 (C-8)、55.9 (C-9)、166.7 (C-11)、55.2 (C-12)、166.2 (C-14)、40.1 (C-15)、126.4 (C-16)、130.7 (C-17, C-21)、114.9 (C-18, C-20)、156.0 (C-19)。以上数据与文献[7]对比基本一致,故确定为cyclo-(L-Tyr-L-Trp)。
化合物2:淡黄色固体,ESI-MS显示准分子离子峰m/z 274 [M+H]+。1H-NMR (600 MHz, DMSO)发现其与化合物1一样有吲哚环的特征信号δH 7.09 (1H, s, H-2),7.52 (1H, d, J = 7.9 Hz, H-4),6.99 (1H, t, J = 7.8 Hz, H-5),7.02 (1H, t, J = 7.8 Hz, H-6),7.30 (1H, d, J = 7.8 Hz, H-7);3个活泼氨基质子信号δH 10.87 (1H, s, NH-1),δH 8.30 (1H, m, NH-10),7.85 (1H, d, J = 2.9 Hz, NH-13);两组亚甲基质子信号δH 3.21 (1H, m, H-8),3.13 (1H, m, H-8),δH 3.65 (1H, m, H-15),3.05 (1H, m, H-15),两个次甲基质子信号δH 4.87 (1H, m, H-9),4.00 (1H, m, H-12)。13C-NMR (150 MHz, DMSO)结合DEPT谱表明其有14个碳信号,2个酮羰基碳δC 167.2,165.7,8个芳香碳,2个亚甲基碳δC 63.0,30.3和2个次甲基碳δC 57.3,55.5。对其碳信号进行归属:δC 127.6 (C-2)、111.2 (C-3)、136.0 (C-3a)、118.6 (C-4)、120.8 (C-5)、124.0 (C-6)、118.3 (C-7)、109.0 (C-7a)、30.3 (C-8)、57.3 (C-9)、167.2 (C-11)、55.5 (C-12)、165.7 (C-14)、63.0 (C-15)。以上数据与文献[8]对比基本一致,故确定为cyclo-(L-Trp-L-Ser)。
化合物3:白色固体,ESI-MS显示准分子离子峰m/z 261 [M+H]+。1H-NMR (600 MHz, DMSO)提示有2个活泼氢质子信号δH 7.87 (1H, s, NH-8),9.22 (1H, s, OH-4’);1组对位二取代的苯环芳香质子信号δH 7.04 (2H, d, J = 8.2 Hz, H-2’, H-6’),6.63 (2H, d, J = 8.2 Hz, H-3’, H-5’);2个次甲基质子信号δH 4.24 (1H, t, J = 8.2 Hz, H-6),4.03 (1H, dd, J = 9.9, 2.9 Hz, H-9);4组亚甲基质子信号δH 3.42 (1H, m, H-3),3.24 (1H, m, H-3),1.73 (2H, m, H2-4),2.00 (1H, m, H-5),1.41 (1H, m, H-5),2.93 (2H, m, H2-10)。13C-NMR (150 MHz, DMSO)结合DEPT谱表明其有14个碳信号,2个酮羰基碳δC 168.9,165.1,6个芳香碳,2个次甲基碳δC 58.4,56.0以及4个亚甲基碳δC 44.6,34.7,27.8,21.9。对其碳信号进行归属:δC 165.1 (C-1)、44.6 (C-3)、21.9 (C-4)、27.8 (C-5)、58.4 (C-6)、168.9 (C-7)、56.0 (C-9)、34.7 (C-10)、127.0 (C-1’)、130.8 (C-2’, C-6’)、114.8 (C-3’, C-5’)、155.9 (C-4’)。以上数据与文献[9]对比基本一致,故确定为cyclo-(D-Tyr-D-Pro)。
化合物4:白色固体,ESI-MS显示准分子离子峰m/z 311 [M+H]+。1H-NMR (600 MHz, DMSO)显示3个活泼氢质子信号δH 9.30 (1H, s, OH-11),7.84 (2H, t, J = 2.9 Hz, NH-1, NH-4);9个芳香区质子信号:4个归为1组对位二取代苯环δH 6.85 (2H, d, J = 8.5 Hz, H-9, H-13),6.65 (2H, t, J = 8.5 Hz, H-10, H-12),5个归为1组单取代苯环δH 7.20 (1H, t, J = 7.6 Hz, H-18),7.04 (2H, d, J = 6.9 Hz, H-16, H-20),7.28 (2H, t, J = 7.6 Hz, H-17, H-19);2组亚甲基质子信号δH 2.58 (1H, dd, J = 13.6, 5.0 Hz, H-7),2.20 (1H, d, J = 6.5 Hz, H-7),2.19 (2H, dd, J = 13.6, 6.5 Hz, H2-14),2个次甲基质子信号δH 3.95 (1H, m, H-3),3.90 (1H, m, H-6)。13C-NMR (150 MHz, DMSO)结合DEPT谱显示其有18个碳信号,2个酮羰基碳δC 166.2,166.2,12个芳香碳,2个亚甲基碳δC 40.1,38.5和2个次甲基碳δC 55.7,55.4。对其碳信号进行归属:δC 166.2 (C-2)、55.7 (C-3)、166.3 (C-5)、55.4 (C-6)、40.1 (C-7)、126.5 (C-8)、130.8 (C-9, C-13)、115.0 (C-10, C-12)、156.1 (C-11)、38.5 (C-14)、136.7 (C-15)、129.7 (C-16, C-20)、128.2 (C-17, C-19)、126.4 (C-18)。以上数据与文献[10]对比基本一致,故确定为cyclo-(L-Tyr-L-Phe)。
化合物5:白色固体,ESI-MS显示准分子离子峰m/z 277 [M+H]+。1H-NMR (600 MHz, DMSO)显示3个活泼氢质子信号δH 9.22 (1H, s, OH-4’),8.02 (2H, dd, J = 5.6, 2.5 Hz, NH-1, NH-4);4个芳香质子信号δH 6.90 (2H, d, J = 8.2 Hz, H-2’, H-6’),6.64 (2H, d, J = 8.2 Hz, H-3’, H-5’),提示分子中有1个对位二取代苯环;3个次甲基质子信号δH 4.06 (1H, q, J = 3.3 Hz, H-3),3.44 (1H, m, H-6),1.43 (1H, ov, H-8),2个亚甲基质子信号δH 1.43 (1H, m, H-7),1.23 (1H, m, H-7),2.69 (1H, q, J = 13.6, 4.8 Hz, H-11),3.01 (1H, q, J = 13.7, 3.7 Hz, H-11)以及2个末端甲基质子信号δH 0.63 (6H, ov, H3-9, H3-10)。13C-NMR (150 MHz, DMSO)结合DEPT谱显示其有15个碳信号,2个酮羰基碳δC 166.2,167.4,6个芳香碳,2个亚甲基碳δC 43.7,37.7,3个次甲基碳δC 55.7,52.3,21.4以及2个甲基碳δC 22.9,22.8。对其碳信号进行归属:δC 166.2 (C-2)、55.7 (C-3)、167.4 (C-5)、52.3 (C-6)、43.7 (C-7)、21.4 (C-8)、22.9 (C-9)、22.8 (C-10)、37.7 (C-11)、125.8 (C-1’)、131.2 (C-2’, C-6’)、114.8 (C-3’, C-5’)、156.4 (C-4’)。以上数据与文献[11]对比基本一致,故确定为cyclo-(L-Tyr-L-Leu)。
化合物6:白色固体,ESI-MS显示准分子离子峰m/z 259 [M+Na]+。1H-NMR (600 MHz, DMSO)显示有2个活泼氢质子信号δH 5.05 (1H, d, J = 4.5 Hz, OH-4)和4.39 (1H, q, J = 4.0 Hz, OH-13),3个甲基质子信号δH 0.88 (3H, d, J = 6.8 Hz, H3-12),0.98 (3H, s, H3-14)和δH 1.05 (3H, s, H3-15),5对亚甲基质子信号δH 2.14 (1H, m, H-3),1.23 (1H, m, H-3),1.74 (1H, m, H-9),1.60 (1H, m, H-9),1.44 (3H, m, H2-10, H-11),1.32 (1H, d, J = 10.4 Hz, H-11),3.95 (2H, m, H2-13),3个次甲基质子信号δH 1.68 (1H, m, H-2),4.57 (1H, m, H-4),1.77(1H, m, H-8)。13C-NMR (150 MHz, DMSO)结合DEPT谱共显示有15个碳信号,包括4个季碳δC 52.1,150.1,136.7,39.8;3个次甲基碳δC 35.2,69.8,46.4;5个亚甲基碳δC 42.4,23.8,28.7,36.4,57.1以及3个甲基碳δC 13.7,29.1,24.4。对其碳信号进行归属:δC 52.1 (C-1)、35.2 (C-2)、42.4 (C-3)、68.9 (C-4)、150.1 (C-5)、136.7 (C-6)、39.8 (C-7)、46.4 (C-8)、23.8 (C-9)、28.7 (C-10)、36.4 (C-11)、13.7 (C-12)、57.1 (C-13)、29.1 (C-14)、24.4 (C-15)。以上数据与文献[12]对比基本一致,故确定为albaflavenol B。
化合物7:白色结晶固体,ESI-MS显示准分子离子峰m/z 268 [M+H]+。1H-NMR (600 MHz, DMSO)可看出其有13个氢信号,包括5个活泼氢质子信号δH 3.56 (2H, m, NH2),5.49 (1H, s, OH-5’),5.36 (1H, t, J = 4.9 Hz, OH-2’)和5.23 (1H, s, OH-3’),4个连氧次甲基质子信号δH 4.56 (1H, s, H-2’),4.14 (1H, s, H-3’),3.96 (1H, m, H-4’)和5.90 (1H, d, J = 5.8 Hz, H-1’),1组亚甲基信号δH 3.66 (2H, m, H2-5’)以及2个低场区的烯氢质子信号δH 8.37 (1H, s, H-8)和8.21 (1H, s, H-2)。13C-NMR (150 MHz, DMSO)显示其共有10个碳信号,结合DEPT谱可推测有3个芳香季碳δC 149.9,119.8,154.3,2个连氮的芳香次甲基碳δC 151.7,138.6,4个次甲基碳δC 87.8,73.5,70.5,85.7,1个亚甲基碳δC 61.5。对其碳信号进行归属:δC 151.7 (C-2)、149.9 (C-4)、119.8 (C-5)、154.3 (C-6)、138.6 (C-8)、87.8 (C-1’)、73.5 (C-2’)、70.5 (C-3’)、85.7 (C-4’)、61.5 (C-5’)。以上数据与文献[13]对比基本吻合,故确定为β-adenosine。
化合物8:绿色无定型固体,ESI-MS显示准分子离子峰m/z 297 [M+H]+。1H-NMR (600 MHz, MeOD)显示有12个氢信号,包括1个活泼氢质子信号δH 8.37 (1H, s, H-11),1个甲氧基质子信号δH 3.79 (3H, s),1个甲基质子信号δH 1.25 (3H, d, J = 6.4 Hz, H3-13),3个低场区的芳香氢质子信号δH 8.31 (1H, d, J = 7.8 Hz, H-4),6.92 (1H, t, J = 8.0 Hz, H-5)和7.65 (1H, d, J = 8.0 Hz, H-6),以及2个次甲基质子信号δH 4.74 (1H, d, J = 3.2 Hz, H-8)和4.40 (1H, m, H-12),后与文献[14]对比发现其有4个活泼氢质子信号没有显示出来,而根据相关化学位移可确定其是同一个已知化合物。13C-NMR (150 MHz, MeOD)显示有13个碳信号,结合DEPT谱可推测有6个芳香碳δC 123.4,119.4,126.2,128.2,152.5,115.6,2个羰基碳δC 171.8,172.4,而δC 162.1为醛基碳,2个次甲基碳δC 59.4,68.4,1个甲基碳δC 20.5,以及1个甲氧基碳δC 52.9。对其碳信号进行归属:δC 115.6 (C-1)、152.5 (C-2)、128.2 (C-3)、126.2 (C-4)、119.4 (C-5)、123.4 (C-6)、171.8 (C-7)、59.4 (C-8)、172.4 (C-9)、52.9 (C-10)、162.1 (C-11)、68.4 (C-12)、20.5 (C-13)。以上数据与文献[14]对比基本吻合,故确定为N-formylantimyic acid methyl ester。
化合物9:白色粉末状固体,ESI-MS显示准分子离子峰m/z 499 [M+H]+。1H-NMR (600 MHz, DMSO)显示有19个氢信号:δH 8.20 (1H, s, H-11),6.82 (1H, s, H-10),6.32 (1H, dd, J = 10.5, 1.3 Hz, H-3),5.01 (1H, m, H-7),2.99 (1H, dd, J = 2.5, 15.8 Hz, H-8),2.80 (1H, dd, J = 10.3, 15.6 Hz, H-8),2.58 (1H, m, H-4),1.66 (1H, m, H-5),1.65 (3H, s, 2-Me),1.27 (1H, m, H-6),1.25 (1H, m, H-5),1.06 (3H, d, J = 6.5 Hz, 4-Me)和0.95 (3H, d, J = 5.9 Hz, 6-Me)。而13C-NMR (150 MHz, DMSO)结合DEPT谱显示只有14个碳信号:δC 166.0 (C-1),126.8 (C-2),147.4 (C-3),30.7 (C-4),37.6 (C-5),35.1 (C-6),74.5 (C-7),24.0 (C-8),149.4 (C-9),122.9 (C-10),151.3 (C-11),12.7 (2-Me),21.1 (4-Me)和16.2 (6-Me),说明这个化合物可能是一个具有对称结构的二聚体,通过与文献[15]中化合物conglobatin A对比后发现两者波谱数据完全吻合,故最终确定为conglobatin A。
3. 讨论
自上世纪发现青霉素以来,微生物中活性次生代谢产物一直是药物先导化合物的重要来源之一,据统计1940年—2019年间,科学家从微生物中开发出293种治疗不同疾病的临床药物[16]。但随着研究的深入,很多微生物及其次生代谢产物存在被重复开发和提取分离的问题,加之多重耐药性的产生,迫使人们需要开拓新的制造药物的微生物来源[5-6],而其中极地微生物资源是珍贵而特殊的。来自极地海洋等特殊生态环境的生物往往具有比陆地生物更为丰富的代谢途径和功能基因簇,增加了产生结构新颖且功能独特的次生代谢物的可能性。极地生物以微生物和一些能适应极端条件的海洋生物为主,然而与已报道的大量极地微生物相比,鲜有微生物活性天然产物相关研究报道,因此,极地微生物极具研究价值[17-18]。
笔者以一株采自北极海域海绵共附生放线菌Streptomyces sp. LHW11-07为研究对象,从其发酵浸膏中分离得到9个单体化合物1~9,包括环二肽化合物1~5,倍半萜化合物6,核苷类化合物7,以及两个其他结构类型化合物8和9,其中化合物1和2是首次分离于Streptomyces放线菌,而这些化合物的生物活性还有待进一步探究;本研究进一步丰富了该属放线菌的化学多样性,同时,为高值化开发利用极地微生物这一国家战略资源提供了物质基础和理论依据。
据文献报道,化合物1对所测试的病原性细菌和真菌均具有一定的对抗作用[19],化合物2测试了4种肿瘤细胞均无明显的细胞毒性[20],化合物3对海胆Strongylocentrotus intermedius胚胎具有细胞毒活性[9],化合物5具有抗炎活性并对H1N1和RSV病毒有一定的杀伤作用[21],化合物7作为一种内源性嘌呤核苷,具有降低血压、抑制血小板聚焦、舒张血管、减慢心律等生理活性[22],而化合物9可抑制癌细胞株的增殖,在体外对Trypanosoma brucei brucei GUTat 3.1表现出抗锥虫体活性等[23]。
-
[1] WANG C, YE Y, SUN W, et al. Red blood cells for glucose-responsive insulin delivery[J]. Adv Mater,2017,29(18):2017May;29(18). [2] KROLL A V, FANG R H, ZHANG L F. Biointerfacing and applications of cell membrane-coated nanoparticles[J]. Bioconjug Chem,2017,28(1):23-32. doi: 10.1021/acs.bioconjchem.6b00569 [3] ZHAI Y H, SU J H, RAN W, et al. Preparation and application of cell membrane-camouflaged nanoparticles for cancer therapy[J]. Theranostics,2017,7(10):2575-2592. doi: 10.7150/thno.20118 [4] WEI X L, GAO J, FANG R H, et al. Nanoparticles camouflaged in platelet membrane coating as an antibody decoy for the treatment of immune thrombocytopenia[J]. Biomaterials,2016,111:116-123. doi: 10.1016/j.biomaterials.2016.10.003 [5] KANG T, ZHU Q Q, WEI D, et al. Nanoparticles coated with neutrophil membranes can effectively treat cancer metastasis[J]. ACS Nano,2017,11(2):1397-1411. doi: 10.1021/acsnano.6b06477 [6] YANG R, XU J, XU L G, et al. Cancer cell membrane-coated adjuvant nanoparticles with mannose modification for effective anticancer vaccination[J]. ACS Nano,2018,12(6):5121-5129. doi: 10.1021/acsnano.7b09041 [7] RAO L, HE Z B, MENG Q F, et al. Effective cancer targeting and imaging using macrophage membrane-camouflaged upconversion nanoparticles[J]. J Biomed Mater Res A,2017,105(2):521-530. doi: 10.1002/jbm.a.35927 [8] VIJAYAN V, UTHAMAN S, PARK I K. Cell membrane coated nanoparticles: an emerging biomimetic nanoplatform for targeted bioimaging and therapy[J]. Adv Exp Med Biol,2018,1064:45-59. [9] MATHIYAZHAKAN M, WIRAJA C, XU C J. A concise review of gold nanoparticles-based photo-responsive liposomes for controlled drug delivery[J]. Nanomicro Lett,2018,10(1):10. [10] GAO C Y, LIN Z H, JURADO-SÁNCHEZ B, et al. Stem cell membrane-coated nanogels for highly efficient in vivo tumor targeted drug delivery[J]. Small,2016,12(30):4056-4062. doi: 10.1002/smll.201600624 [11] CHEN W, OUYANG J, YI X, et al. Black phosphorus nanosheets as a neuroprotective nanomedicine for neurodegenerative disorder therapy[J]. Adv Mater,2018,30(3):2018Jan;30(3). [12] LIU T, SHI C Z, DUAN L Q, et al. A highly hemocompatible erythrocyte membrane-coated ultrasmall selenium nanosystem for simultaneous cancer radiosensitization and precise antiangiogenesis[J]. J Mater Chem B,2018,6(29):4756-4764. doi: 10.1039/C8TB01398E [13] HE W P, FRUEH J, WU Z W, et al. Leucocyte membrane-coated Janus microcapsules for enhanced photothermal cancer treatment[J]. Langmuir,2016,32(15):3637-3644. doi: 10.1021/acs.langmuir.5b04762 [14] RAO L, CAI B, BU L L, et al. Microfluidic electroporation-facilitated synthesis of erythrocyte membrane-coated magnetic nanoparticles for enhanced imaging-guided cancer therapy[J]. ACS Nano,2017,11(4):3496-3505. doi: 10.1021/acsnano.7b00133 [15] FAN Z Y, LI P Y, DENG J J, et al. Cell membrane coating for reducing nanoparticle-induced inflammatory responses to scaffold constructs[J]. Nano Res,2018,11(10):5573-5583. doi: 10.1007/s12274-018-2084-y [16] LI J H, AI Y W, WANG L H, et al. Targeted drug delivery to circulating tumor cells via platelet membrane-functionalized particles[J]. Biomaterials,2016,76:52-65. doi: 10.1016/j.biomaterials.2015.10.046 [17] WANG Y, ZHANG K, QIN X, et al. Biomimetic nanotherapies: red blood cell based core-shell structured nano complexes for atherosclerosis management[J]. Adv Sci (Weinh),2019,6(12):1900172. doi: 10.1002/advs.201900172 [18] HAN X, WANG C, LIU Z. Red blood cells as smart delivery systems[J]. Bioconjug Chem,2018,29(4):852-860. doi: 10.1021/acs.bioconjchem.7b00758 [19] HU C M J, FANG R H, LUK B T, et al. ‘Marker-of-self’ functionalization of nanoscale particles through a top-down cellular membrane coating approach[J]. Nanoscale,2013,5(7):2664-2668. doi: 10.1039/c3nr00015j [20] FANG R H, HU C M J, CHEN K N H, et al. Lipid-insertion enables targeting functionalization of erythrocyte membrane-cloaked nanoparticles[J]. Nanoscale,2013,5(19):8884-8888. doi: 10.1039/c3nr03064d [21] HU C M J, FANG R H, COPP J, et al. A biomimetic nanosponge that absorbs pore-forming toxins[J]. Nat Nanotechnol,2013,8(5):336-340. doi: 10.1038/nnano.2013.54 [22] HU C M J, FANG R H, WANG K C, et al. Nanoparticle biointerfacing by platelet membrane cloaking[J]. Nature,2015,526(7571):118-121. doi: 10.1038/nature15373 [23] WEI X, YING M, DEHAINI D, et al. Nanoparticle Functionalization with Platelet Membrane Enables Multifactored Biological Targeting and Detection of Atherosclerosis. ACS Nano, 2018. 12(1): 109-116. [24] DE ÁVILA B E F, ANGSANTIKUL P, RAMÍREZ-HERRERA D E, et al. Hybrid biomembrane-functionalized nanorobots for concurrent removal of pathogenic bacteria and toxins[J]. Sci Robot,2018,3(18):eaat0485. doi: 10.1126/scirobotics.aat0485 [25] QIAN B Z, POLLARD J W. Macrophage diversity enhances tumor progression and metastasis[J]. Cell,2010,141(1):39-51. doi: 10.1016/j.cell.2010.03.014 [26] CHRISTIE C, MADSEN S J, PENG Q, et al. Macrophages as nanoparticle delivery vectors for photothermal therapy of brain tumors[J]. Ther Deliv,2015,6(3):371-384. doi: 10.4155/tde.14.121 [27] XUAN M J, SHAO J X, DAI L R, et al. Macrophage cell membrane camouflaged mesoporous silica nanocapsules for in vivo cancer therapy[J]. Adv Healthc Mater,2015,4(11):1645-1652. doi: 10.1002/adhm.201500129 [28] XUAN M J, SHAO J X, DAI L R, et al. Macrophage cell membrane camouflaged Au nanoshells for in vivo prolonged circulation life and enhanced cancer photothermal therapy[J]. ACS Appl Mater Interfaces,2016,8(15):9610-9618. doi: 10.1021/acsami.6b00853 [29] ZHANG Y, CAI K M, LI C, et al. Macrophage-membrane-coated nanoparticles for tumor-targeted chemotherapy[J]. Nano Lett,2018,18(3):1908-1915. doi: 10.1021/acs.nanolett.7b05263 [30] LIM K, HYUN Y M, LAMBERT-EMO K, et al. Neutrophil trails guide influenza-specific CD8 + T cells in the airways[J]. Science,2015,349(6252):1055. doi: 10.1126/science.aad0867 [31] GAO J, CHU D F, WANG Z J. Cell membrane-formed nanovesicles for disease-targeted delivery[J]. J Control Release,2016,224:208-216. doi: 10.1016/j.jconrel.2016.01.024 [32] ZHANG Q Z, DEHAINI D, ZHANG Y, et al. Neutrophil membrane-coated nanoparticles inhibit synovial inflammation and alleviate joint damage in inflammatory arthritis[J]. Nat Nanotechnol,2018,13(12):1182-1190. doi: 10.1038/s41565-018-0254-4 [33] WEI X L, ZHANG G, RAN D N, et al. T-cell-mimicking nanoparticles can neutralize HIV infectivity[J]. Adv Mater,2018,30(45):e1802233. doi: 10.1002/adma.201802233 [34] TOLEDANO FURMAN N E, LUPU-HABER Y, BRONSHTEIN T, et al. Reconstructed stem cell nanoghosts: a natural tumor targeting platform[J]. Nano Lett,2013,13(7):3248-3255. doi: 10.1021/nl401376w [35] CAO B R, YANG M Y, ZHU Y, et al. Stem cells loaded with nanoparticles as a drug carrier for in vivo breast cancer therapy[J]. Adv Mater,2014,26(27):4627-4631. doi: 10.1002/adma.201401550 [36] ANDERSON J M, RODRIGUEZ A, CHANG D T. Foreign body reaction to biomaterials[J]. Semin Immunol,2008,20(2):86-100. doi: 10.1016/j.smim.2007.11.004 [37] RAO L, BU L L, CAI B, et al. Cancer cell membrane-coated upconversion nanoprobes for highly specific tumor imaging[J]. Adv Mater,2016,28(18):3460-3466. doi: 10.1002/adma.201506086 [38] CHEN Z, ZHAO P F, LUO Z Y, et al. Cancer cell membrane-biomimetic nanoparticles for homologous-targeting dual-modal imaging and photothermal therapy[J]. ACS Nano,2016,10(11):10049-10057. doi: 10.1021/acsnano.6b04695 [39] SUN H P, SU J H, MENG Q S, et al. Cancer-cell-biomimetic nanoparticles for targeted therapy of homotypic tumors[J]. Adv Mater,2016,28(43):9581-9588. doi: 10.1002/adma.201602173 [40] HOSSEINIDOUST Z, MOSTAGHACI B, YASA O, et al. Bioengineered and biohybrid bacteria-based systems for drug delivery[J]. Adv Drug Deliv Rev, 2016, 106(Pt A): 27-44. [41] POETSCH A, WOLTERS D. Bacterial membrane proteomics[J]. Proteomics,2008,8(19):4100-4122. doi: 10.1002/pmic.200800273 [42] GAO W W, FANG R H, THAMPHIWATANA S, et al. Modulating antibacterial immunity via bacterial membrane-coated nanoparticles[J]. Nano Lett,2015,15(2):1403-1409. doi: 10.1021/nl504798g [43] ZHANG A N, WU W, ZHANG C, et al. A versatile bacterial membrane-binding chimeric peptide with enhanced photodynamic antimicrobial activity[J]. J Mater Chem B,2019,7(7):1087-1095. doi: 10.1039/C8TB03094D -

 点击查看大图
点击查看大图
计量
- 文章访问数: 8362
- HTML全文浏览量: 15574
- PDF下载量: 272
- 被引次数: 0




 下载:
下载:

 下载:
下载:
