-
光动力治疗(PDT)基于光辐照聚集光敏剂的肿瘤组织,由光敏剂诱发光动力反应形成单线态氧(1O2)等活性氧(ROS),通过对肿瘤细胞和肿瘤血管的直接杀伤及激活机体系统免疫反应等多种机制发挥抗肿瘤作用[1-3]。二氢卟吩及菌绿素类光敏剂是PDT新药研究的热点[4-8]。其中,已获批上市的代表药物有他拉泊芬(talaporfin)和帕利泊芬(padeliporfin)等[9, 10]。
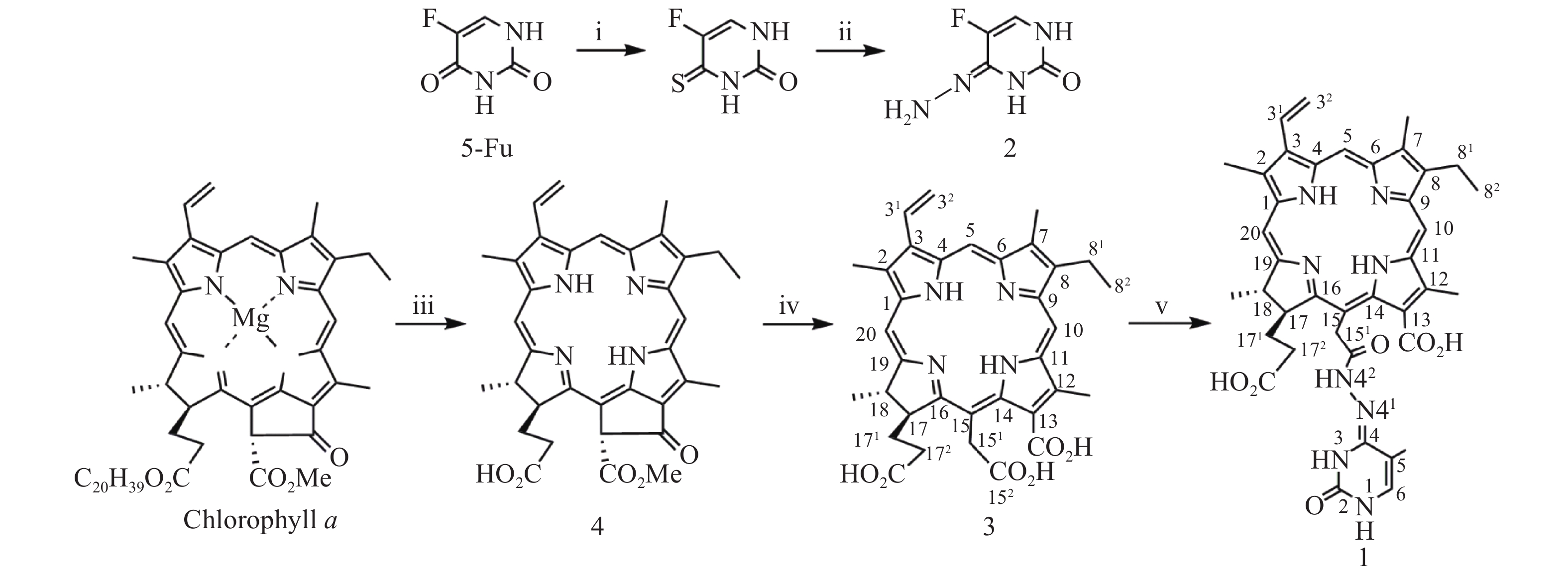
光敏剂作为结构非特异性药物,存在缺乏肿瘤靶向性摄入和明确的作用药靶等缺陷。此外,PDT受制于局部治疗,对浸润较深的肿瘤组织,及已发生转移的肿瘤疗效有限。目前,PDT和化疗联用是克服上述缺陷,提高PDT疗效最为普遍和有效的策略之一。研究表明,抗代谢化疗药物氟尿嘧啶(5-Fu)与PDT联用具有协同抗肿瘤作用[11-13]。据此,我们设想利用在肿瘤微环境下能响应性断裂的连接基团(linker)将光敏剂与化疗药物偶联,希望实现二者在肿瘤组织的靶向释放,从而发挥其PDT和化疗协同抗肿瘤作用。酰腙键是酸敏感化学键,常被用来连接载体,以药物制备智能药物载体。这种药物载体到达肿瘤细胞的内涵体或溶酶体中时,会发生酸性水解将药物有效释放出来。因此,本文针对肿瘤微环境呈弱酸性的特点,采用药物化学最经典的前药设计策略,以脱镁叶绿素a(Phephorbide a)粗提物经酸碱降解制得的二氢卟吩e6(3)[14]为先导光敏剂,通过其152-羧基与抗肿瘤药物5-Fu以酸敏感酰腙键连接,设计合成pH响应型光化疗协同抗肿瘤光敏剂二氢卟吩e6-偕氟尿嘧啶(1),并考察其体外PDT抗肿瘤活性和pH响应性5-Fu释放,及其对黑色素瘤B16-F10和肝癌HepG2细胞的光动力抗癌活性及其作用机制,以期获得高效、低毒的PDT治癌药物候选药物,合成路线见图1。
-
用Bruker MSL-600型核磁共振仪测定1H NMR,CD3OD为溶剂;用API-3000 LC-MS型电喷雾质谱仪测定质谱(ESI-MS);用岛津UV-160型紫外分光光度计测定UV吸收谱;用日立F-7000荧光分光光度计测定荧光发射谱;用Shimazu LC-20AD HPLC仪测定化合物1的相对纯度及其5-Fu的体外释放。色谱柱型号为Waters Xterra C18柱,流动相:乙腈-0.3%乙酸水溶液(80 : 20);流速:1.0 ml/min;检测波长:400 nm(化合物1的相对纯度)或254 nm(5-Fu释放);柱温:30 ℃;进样量:20 μl。柱色谱分离用TELEDYNE ISCO的快速制备色谱Combi Flash@Rf+仪,硅胶H作为固定相。PDT抗癌活性测试使用BWT半导体激光仪(北京凯普林,波长为660 nm);用流式细胞仪(BD Accuri C6,美国)(激发波长:488 nm,发射波长:525 nm)检测受试肿瘤细胞样品的ROS水平、细胞凋亡率和细胞周期阻滞。
二氢卟吩e6(3)按照文献[14]的方法制备;其它实验用材料和化学试剂均为市售商品。
-
取氟尿嘧啶(0.2 g,1.563 mmol)溶于无水吡啶(10 ml),加入五硫化二磷(0.298 g,1.563 mmol),加热回流12 h。反应完毕,减压回收溶剂,残物加乙酸乙酯溶解(100 ml),用0.1 mol/L HCl洗涤(50 ml×2),无水Na2SO4干燥,减压除去溶剂得4-硫代-5-氟尿嘧啶粗品。上述4-硫代-5-氟尿嘧啶粗品加甲醇(10 ml)溶解,于0 ℃下滴加N2H4·H2O(0.316 g,6.252 mmol),室温继续搅拌2 h。反应完毕,减压抽滤,P2O5真空干燥得固体化合物5-氟尿嘧啶-4-腙(2)中间体,直接用于下步反应。取二氢卟吩e6(0.1 g,0.168 mmol)溶于无水DMF(10 ml),加1-乙基-(3-二甲氨基丙基)碳二亚胺盐酸盐(EDC·HCl)(0.035 g,0.183 mmol),室温搅拌反应6 h后再加入中间体2(0.031 g,0.218 mmol),继续搅拌36 h。反应完毕,反应液加入10倍体积量乙酸乙酯,饱和NaCl水溶液洗涤(50 ml×3),无水Na2SO4干燥,减压回收溶剂所得固体经快速制备色谱梯度洗脱分离纯化(流动相为二氯甲烷/甲醇/甲酸=15∶1∶0.1~8∶1∶0.1)得黑色固体1纯品0.048 g,产率39.6%。UV-vis λmax (MeOH, nm) (ε, M−1cm−1):660 (3.15×104), 510 (0.82×104), 402 (8.13×104)。1H-NMR (600 MHz, CD3OD, δ, ppm): 9.79 (s, 1H, 10-CH), 9.73 (s, 1H, 5-CH), 9.07 (s, 1H, 20-CH), 8.19 (dd, J = 18.0, 12.0 Hz, 1H, 31-CH), 7.29 (s, 1H, 5-Fu的6-CH), 6.38 (d, J = 18.0 Hz, 1H, 32-CHB), 6.15 (d, J = 12.0 Hz, 1H, 32-CHA), 5.35 (s, 2H, 151-CH2), 4.65 (m, 2H, 17-CH和18-CH), 3.84 (q, J = 7.5 Hz, 2H, 81-CH2), 3.63 (s, 3H, 12-CH3), 3.53 (s, 3H, 2-CH3), 3.30 (s, 3H, 7-CH3), 2.3~2.0 (m,4H , 171-CH2 和172-CH2), 1.76 (m, 6H¸ 18-CH3和82-CH3)。MS (ESI+) m/z: 723.63 (M+H)+ (100%)。元素分析(C38H39N8O6F,%)计算值:C 63.16, H 5.40, N 15.48;实测值:C 63.34, H 5.38, N 15.43。HPLC测定纯度:95.2%。
-
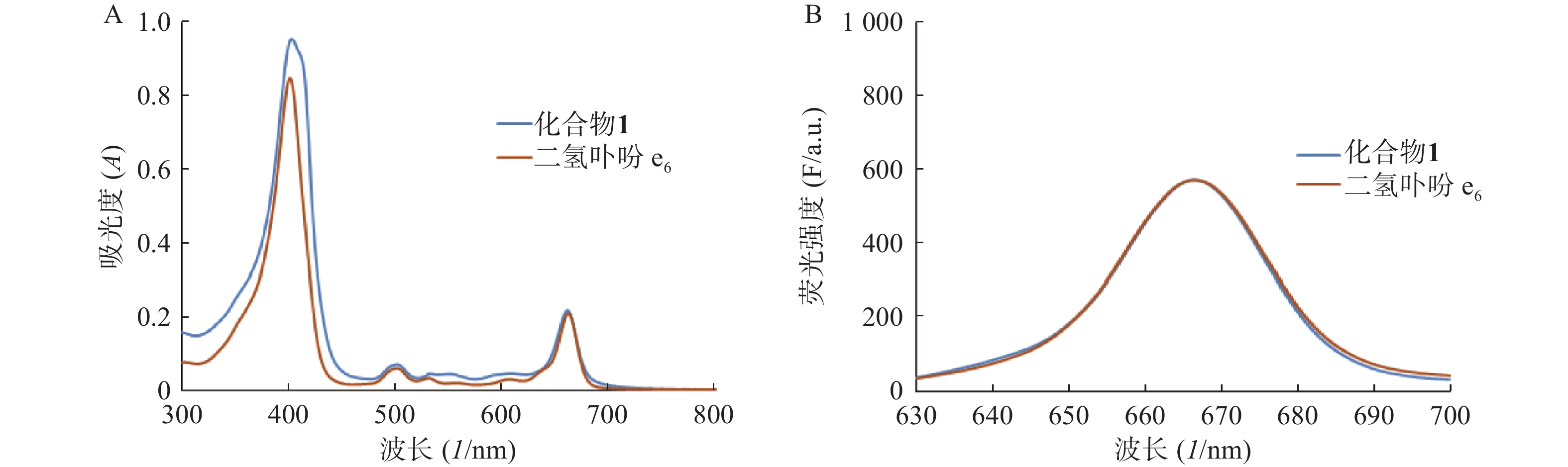
分别测定目标化合物1及其先导化合物二氢卟吩e6(3)的甲醇溶液(10 μmol/L)在300~800 nm处的紫外吸收谱和激发波长为400 nm的荧光发射光谱,结果见图2。
-
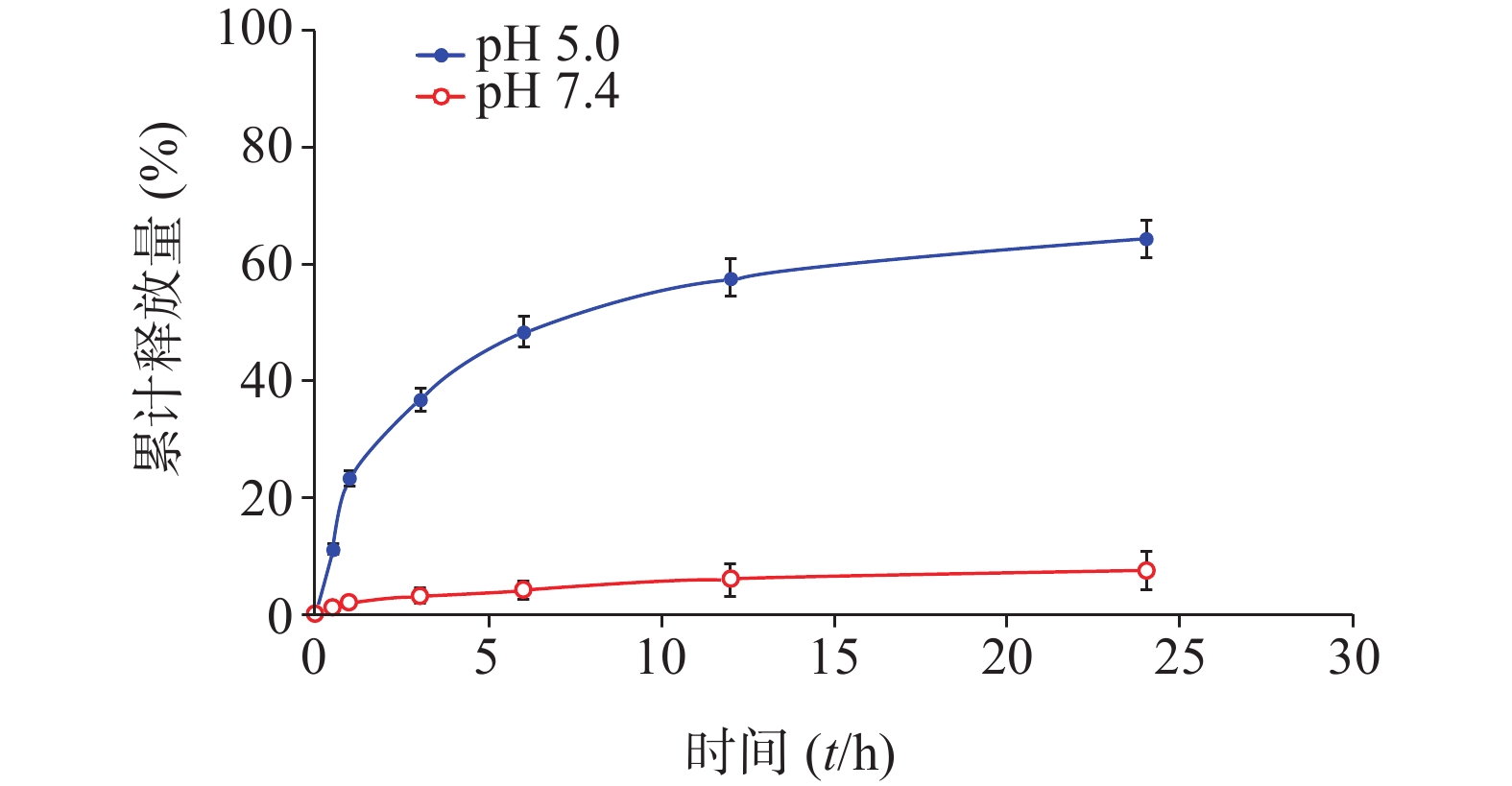
分别配制浓度为50 μmol/L的化合物1的HOAc-NaOAc缓冲液(pH 5.0)和PBS溶液(10 ml),并于0.5、1.0、3.0、6.0、12、24 h时分别取样(500 μl)。其中,HOAc-NaOAc缓冲液(pH 5.0)组取样液用0.1 mol/L氢氧化钠水溶液迅速调节pH值至7.4。每份取样液加PBS稀释至原溶液1/3浓度,微孔滤膜(孔径0.22 μm)过滤,HPLC进样检测;实验重复3次。根据5-Fu的HPLC峰面积-浓度标准曲线分析计算,绘制目标化合物1于弱酸(pH 5.0)中的5-Fu体外释放量-时间曲线,结果见图3。
-
参照文献[6-8]的方法,每孔5×103个B16-F10细胞或HepG2细胞悬液(100 μl)接种于96孔板上,加入等体积上述细胞培养液孵育24 h;更换含不同浓度待测物的培养液(DMSO浓度小于1%,100 μl),继续避光孵育48 h;再更换含10%(V/V)CCK-8(Beyotime,中国)的RPMI 1640基础培养基(100 μl),继续培养1.5 h,然后用Varioskan Flash全波长酶标仪(Thermo)于波长450 nm处测定每孔的吸光度值,计算各浓度对应的细胞存活率,并拟合得到待测物的肿瘤细胞半数抑制浓度即IC50值。
-
每孔5×103个B16-F10细胞或HepG2细胞悬液(100 μl)接种于96孔板上,加入等体积细胞培养液孵育24 h;更换含不同浓度待测物的细胞培养液(DMSO浓度小于1%,100 μl),继续避光孵育24 h;再更换新鲜培养液(100 μl),以波长为660 nm的激光辐照受试细胞样品(光照剂量为10 J/cm2),继续孵育24 h。最后按“2.3.2”项下CCK-8法测定各待测物的肿瘤细胞IC50值。
-
以临床光敏药物他拉泊芬为阳性对照,化合物1及其先导化合物3对肿瘤细胞株的体外PDT抗癌活性结果见表1。
表 1 目标化合物1的体外光动力抗癌活性(IC50,μmol/L)
化合物 B16-F10细胞 暗毒/光毒比 HepG2细胞 暗毒/光毒比 暗毒性 光毒性 暗毒性 光毒性 化合物 1 46.84±8.46*, ΔΔΔ 0.73±0.16**, ΔΔΔ 64.2 50.80±6.45**, #, ΔΔΔ 0.90±0.22**, ΔΔΔ 56.4 二氢卟吩e6 69.72±4.69 3.36±0.59 20.8 70.38±10.9 2.75±0.41 25.6 他拉泊芬 254.8±18.8 11.31±3.88 22.5 176.4±28.4 15.47±5.07 11.4 5-Fu 35.80±6.68 NTa − 39.16±2.7 NTa − NTa:未测定;*P < 0.05,**P < 0.01,与二氢卟吩 e6组比较;#P < 0.05,与5-Fu组比较;ΔΔΔP < 0.001,与他拉泊芬组比较。 -
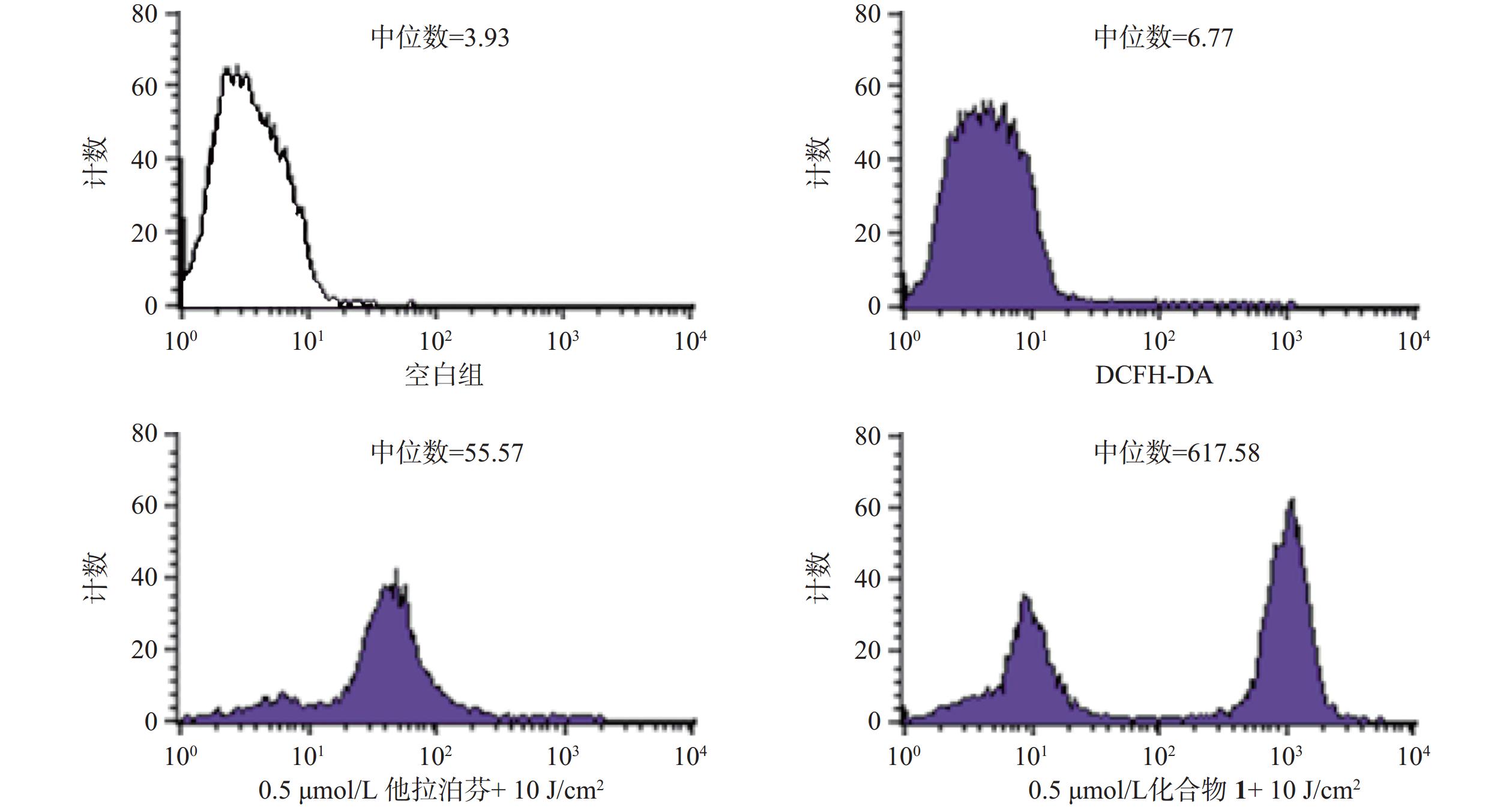
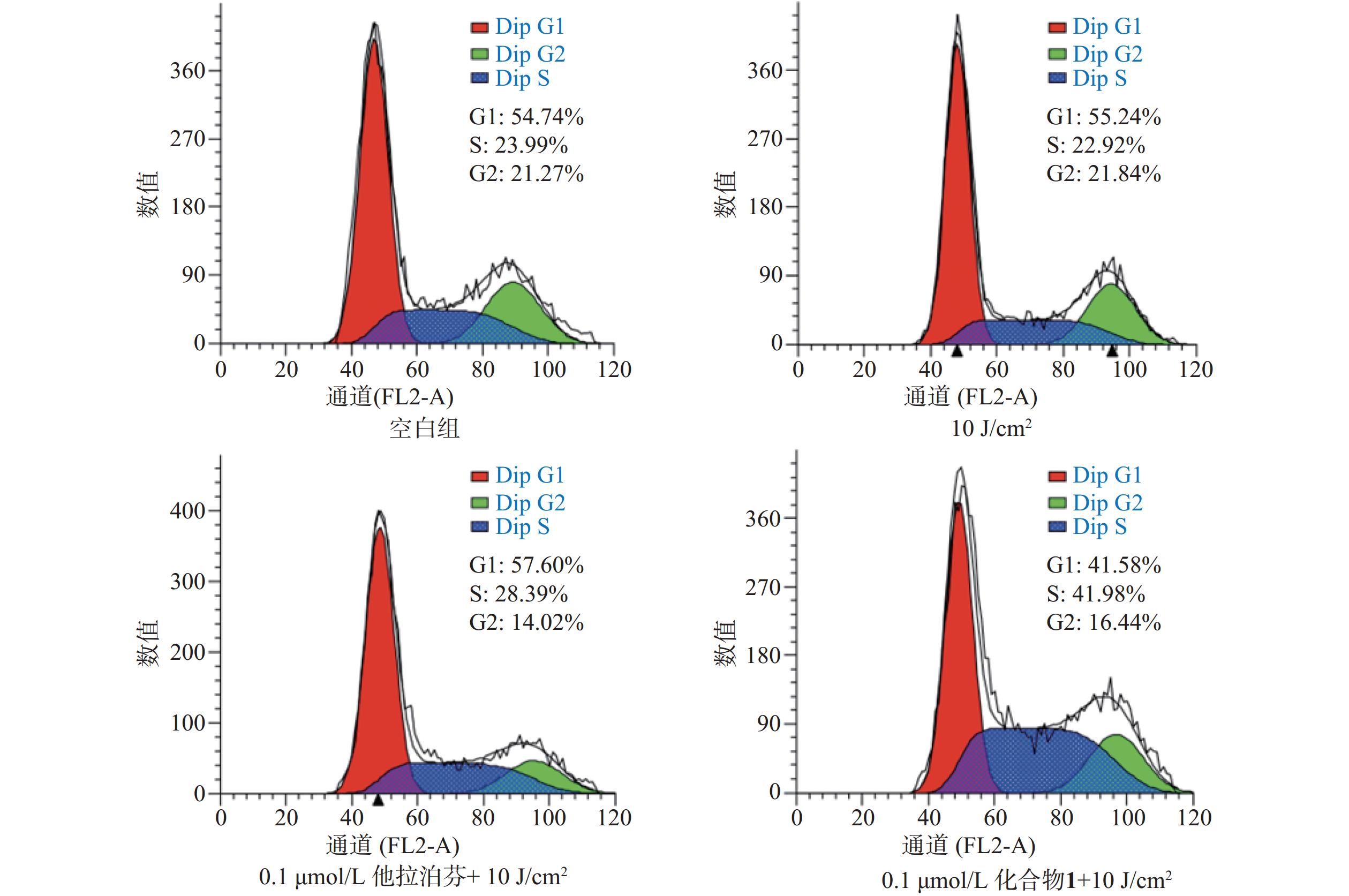
操作步骤如下:a. 每孔3 × 105个B16-F10细胞悬液(2 ml)接种6孔板上,按“2.3.1”项条件避光孵育24 h;b. 分别更换含一定浓度化合物1或他拉泊芬的新鲜培养液(DMSO浓度小于1%,2 ml),继续避光孵育24 h;c. 加入10 mmol/L DCFH-DAROS荧光检测探针(Beyotime,1.5 μl),吹打混匀,继续避光孵育20 min;d. PBS洗涤3次,再加新鲜培养液(2 ml),以660 nm波长的激光辐照(光剂量10 J/cm2)细胞样品,继续避光孵育20 min;e. 收集每孔细胞样品,用流式细胞仪检测各孔细胞ROS水平,结果见图4。
-
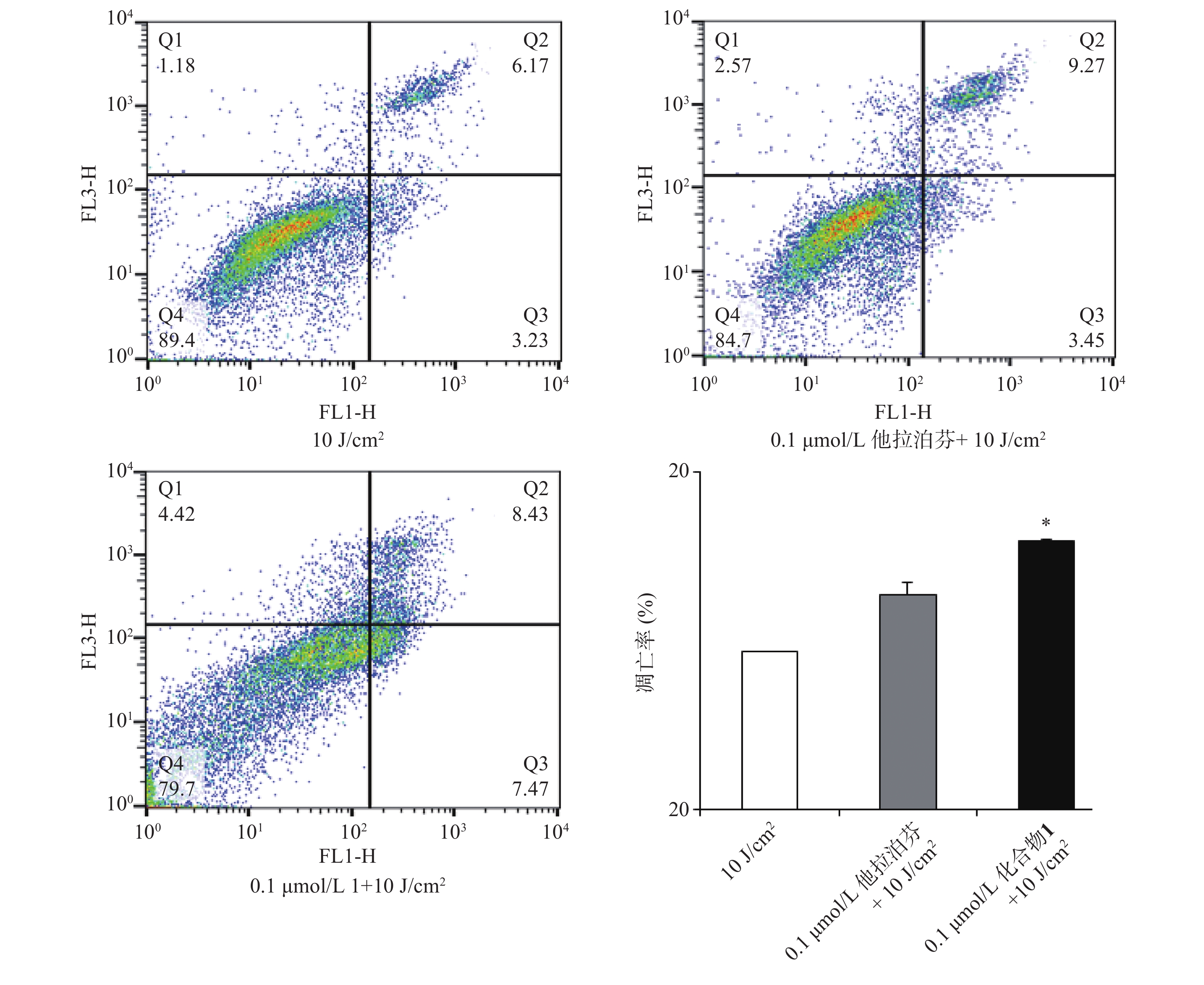
按“2.4”项下操作方法,仅从步骤c开始,更换新鲜培养液(2 ml),用660 nm波长的激光辐照(光剂量10 J/cm2)细胞样品,继续避光孵育20 min;d. 以1 500 r/min离心(5 min)细胞样品,PBS洗涤,再以1 000 r/min离心(5 min)后获取细胞样品;e. 按Annexin V-FITC细胞凋亡检测试剂盒(Beyotime)操作流程操作,结果见图5。
-
按“2.5”项下操作方法,仅在e步骤中,换以细胞周期阻滞检测试剂盒(Beyotime)的操作流程,每份细胞样品中分别加入染色缓冲液(300 µl)、RNase A(6 µl)和碘化丙啶染色液(15 µl),轻轻混匀,避光孵育20 min后,用流式细胞仪进行细胞周期阻滞检测,结果见图6。
-
按文献[14]方法制得的二氢卟吩e6(3)为先导化合物,经1-乙基-(3-二甲氨基丙基)碳二亚胺盐酸盐(EDC·HCl)于无水DMF中催化分子内脱水缩合制得二氢卟吩e6-131,152-酸酐活泼中间体[15],然后直接与中间体2发生酰化反应成功合成得到了光化疗双模抗肿瘤光敏剂二氢卟吩e6-偕氟尿嘧啶(1),反应收率达39.6%,其结构经UV、ESI-MS、1H NMR及元素分析确证。
化合物1在甲醇中最大紫外吸收波长和荧光发射波长(激发波长:400 nm)分别为660 nm和670 nm,与先导物3相一致,表明先导物3以酰腙键偶联5-Fu后,并没有改变其作为光敏剂特有的紫外吸收和荧光发射光谱等光物理特性。此外,化合物1在弱酸(pH 5.0)条件下,能有效释放5-Fu,24 h内累积释放率可达60.3%;但在pH 7.4的条件下较为稳定,24 h内5-Fu累积释放率仅为5%。
体外PDT抗癌活性测试结果显示,化合物1对B16-F10和HepG2细胞株的光毒活性和暗毒/光毒比(治疗指数)均显著优于先导物二氢卟吩e6(3)(P<0.005)和他拉卟吩(P<0.001),其IC50值分别达0.73 μmol/L和0.90 μmol/L。
体外PDT抗癌机制研究提示,化合物1介导的PDT能显著提升B16-F10细胞内ROS水平和诱导B16-F10细胞凋亡,并阻滞肿瘤细胞周期于S期。
总之,二氢卟吩e6-偕氟尿嘧啶(1)具有PDT抗癌活性强、治疗指数(暗毒/光毒比)高且可在肿瘤弱酸环境中有效释放5-Fu等优点,从而实现“单分子”光化疗协同抗肿瘤作用,值得进一步开发研究。
Synthesis and biological activities of chlorin e6-based conjugate of fluorouracil as dual-mode antitumor photosensitizer
-
摘要:
目的 文献报道氟尿嘧啶(5-Fu)与光敏剂联用具有协同抗肿瘤作用,笔者设计合成二氢卟吩e6(化合物 3 )与5-Fu经酰腙键偶联的pH响应性、光化疗双模抗肿瘤光敏剂(化合物 1 ),研究其初步体外光动力抗癌活性及作用机制。 方法 首先,5-Fu与五硫化二磷于吡啶中回流反应形成4-硫代-5-氟尿嘧啶,再和水合肼于甲醇中反应制得5-氟尿嘧啶-4-腙(化合物 2 );然后,将脱镁叶绿素a(化合物 4 )酸碱降解产物 3 经EDC·HCl催化缩合形成二氢卟吩e6-131,152-酸酐中间体后,直接与 2 发生选择性酰化反应,制得目标化合物 1 ,并考察其体外pH响应性5-Fu释放及对黑素瘤B16-F10和肝癌HepG2细胞的光动力抗癌活性和作用机制。 结果 化合物 1 在微酸(pH 5.0)环境中能有效释放5-Fu,24 h累积释放率可达60.3%;其在光照下对黑素瘤B16-F10和肝癌HepG2细胞的半数抑制浓度(IC50)分别为0.73 μmol/L和0.90 μmol/L,均显著优于先导物 3 和上市药物他拉泊芬(talaporfin),且能显著提升肿瘤细胞内活性氧(ROS)水平和诱导肿瘤细胞凋亡,并阻滞肿瘤细胞周期于S期。其结构经紫外、电喷雾质谱、氢谱和元素分析确证。 结论 新型双模抗肿瘤光敏剂化合物 1 具有光动力抗癌活性强、治疗指数(暗毒/光毒比)高,且可在微酸(pH 5.0)环境响应性释放5-Fu等优点,从而实现“单分子”光化疗双重抗肿瘤作用,值得进一步开发研究。 Abstract:Objective To design and synthesize the conjugate (compound 1 ) of chlorin e6 (compound 3 ) with fluorouracil (5-Fu) as novel pH-responsive dual-mode antitumor photosensitizer by acyl hydrazone bond coupling, based on literature reports that combination of 5-Fu and photosensitizer possess synergistic anti-tumor effect, and investigate its photodynamic antitumor activity and mechanism. Methods Lead compound 3 was obtained by alkali degradation with 25% KOH-CH3OH on pheophorbide a (compound 4 ) which was prepared through acid hydrolysis of chlorophyll a in crude chlorophyll extracts from silkworm excrement. Reflux reaction of 5-Fu with P2S5 in pyridine formed crude 4-thio-5-fluorouracil which was followed to react with hydrazine hydrate (N2H4·H2O) in CH3OH to give 5-fluorouracil-4-hydrazone (compound 2 ). Then, treatment of compound 3 i.e. acid alkali degradation product of chlorophyll a in silkworm excrement with EDC·HCl generated its 171- and 152 cyclic anhydride which was followed to directly react with intermediate compound 2 to successfully get title compound 1 . In addition, its pH-responsive 5-Fu release and photodynamic antitumor activity and their mechanisms in vitro were investigated. Results Compound 1 could responsively release 5-Fu at pH 5.0, with a cumulative release rate of 60.3% within 24 h. It exhibited much higher phototoxicity against melanoma B16-F10 and liver cancer HepG2 cells than talaporfin and its precursor compound 3 , with IC50 value being 0.73 μmol/L for B16-F10 cells and 0.90 μmol/L for HepG2 cells, respectively. Upon light irradiation, it also could significantly induce cell apoptosis and intracellular ROS level and block cell cycle in S phase. Its structure was confirmed by UV, 1H-NMR, ESI-MS and elemental analysis data. Conclusion The conjugate compound 1 of compound 3 and 5-Fu has the advantages of strong PDT anticancer activity, high therapeutic index (i.e. dark toxicity/phototoxicity ratio) and responsively release 5-Fu at pH 5.0 etc. which shows “unimolecular” dual antitumor effects of PDT and chemotherapy and is worthy of further research and development. -
Key words:
- synthesis /
- photodynamic therapy /
- photosensitizer /
- chlorin e6 /
- fluorouracil (5-Fu) /
- antitumor
-
山楂为蔷薇科植物山里红Crataegus pinnatifida Bge. var. major N. E. Br. 或山楂 Crataegus pinnati- fida Bge. 的干燥成熟果实,焦山楂为其炒制品[1]。现代药理研究证明,焦山楂抑菌作用强于生山楂,而某些特定菌群与消化功能密切相关[2]。而山楂炒焦后产生新的物质—类黑素,类黑素是在食品热处理过程中形成的。目前,类黑素的抗菌活性已得到证实。大多数类黑素对微生物作用的研究都是在特定的微生物生长培养基中进行的,这些研究表明类黑素可以刺激微生物生长[3],也可以抑制微生物生长[4-5]。肠道菌群与人体健康密切相关,药物和功能食品可能通过调节肠道微生物来改善胃肠功能,帮助消化[6-7]。双歧杆菌和大肠杆菌是典型的有益菌和有害菌,双歧杆菌常被加入酸奶饮品中帮助消化。乙酸是双歧杆菌的主要代谢物质,随着乙酸的增多,pH值降低从而抑制大肠杆菌的生长繁殖。本实验通过研究山楂,焦山楂以及焦山楂炒制过程中产生的类黑素对大肠杆菌、双歧杆菌以及其代谢物乙酸的影响,探究“山楂炒焦长于消食导滞”的作用机制。
1. 材料与方法
1.1 材料
1.1.1 实验仪器
低温培养箱(美墨尔特有限公司,德国),生物安全柜(赛默飞世尔科技公司,美国),高压灭菌锅(三洋公司,日本),纯水机(密理博公司,美国),厌氧罐(北京陆桥技术股份有限公司,北京);紫外可见分光光度计(上海佑科仪器仪表有限公司,上海);7890B型气相色谱仪(安捷伦科技有限公司,美国);HP-FFAP型毛细管柱(货号:19091F-413,安捷伦科技有限公司,美国);GM900型非接触红外测温仪(深圳聚茂源科技有限公司,深圳)。
1.1.2 实验试剂
MRS固体培养基、PYG液体培养基、厌氧产气袋和厌氧指示剂(北京陆桥技术股份有限公司);蛋白胨、酵母粉(英国OXOID公司);乙酸(98.85%,国药集团化学试剂有限公司,中国);其余试剂均为分析纯。
1.1.3 山楂和实验菌株
净山楂饮片(四川同善堂中药饮片有限责任公司,批号:180501);双歧杆菌(GDMCC1.1258)、大肠杆菌(ATCC25922)(中国科学院微生物研究所)。
1.2 方法
1.2.1 焦山楂的炮制
参照2015版《中国药典》一部山楂项下制备焦山楂。取生山楂150 g,中火(380~420)℃炒制10 min,至药材表面呈焦黄或焦褐色,内部颜色加深,并具有焦香气味,取出,常温封存,即得。
1.2.2 生山楂,焦山楂和类黑素浸膏的制备
(1)生山楂和焦山楂浸膏的制备
取生山楂和焦山楂各100 g进行水浸提,料液比为1:15,浸提8 h,浸提2次。生山楂和焦山楂浸提液分别在4 ℃下以3 600 r/min离心10 min,取上层清液各1 000 ml。将500 ml上层清液进行蒸发浓缩至胶状,停止加热,余温使其自然干燥,得生山楂浸膏13.75 g,焦山楂浸膏14.02 g。
(2)焦山楂中类黑素的提取
取焦山楂100 g,按照“1.2.2”项中⑴的方法提取得到1 000 ml上层清液。取500 ml上层清液蒸发浓缩得棕褐色浓缩液50 ml,进行大孔树脂吸附,室温吸附流速1.5 ml/min,60%乙醇作为洗脱剂,洗脱至色谱柱上无棕色为止,收集洗脱液500 ml。洗脱液蒸发浓缩至胶状,停止加热,余温使其自然干燥,得焦山楂类黑素浸膏13.12 g。
(3)类黑素的紫外检测
取类黑素浸膏1 g,蒸馏水溶解定容至100 ml,取10 ml溶液,分别定容至50 ml;因波长420 nm处是类黑素的特征吸收波长,测其特征吸收下的吸光度值,焦山楂类黑素浸膏吸光度值为0.492,说明焦山楂中类黑素提取成功。
1.2.3 山楂炮制品及类黑素对肠道菌群生长繁殖的影响
(1)双歧杆菌测试菌菌液的制备
以接种环自双歧杆菌标准菌种管挑取菌种,划线接种至MRS固体培养基,36 ℃厌氧培养48 h,挑取单菌落接种至PYG液体培养基,36 ℃厌氧培养48 h,以生理盐水调整浓度至1.0麦氏浓度,作为受试菌初始菌液,按10:1浓度加入试验体系。
(2)大肠杆菌测试菌液的制备
以接种环自大肠杆菌标准菌种管挑取菌种,划线接种至LB固体培养基(配方:蛋白胨10 g,酵母粉5 g,氯化钠10 g,琼脂粉15 g,加入1 L蒸馏水,以5 mol/L氢氧化钠调节pH至7.0,121 ℃高压灭菌15 min备用),36 ℃有氧培养24 h,挑取单菌落接种至LB液体培养基(配方:蛋白胨10 g,酵母粉5 g,氯化钠10 g,加入1 L蒸馏水,以5 mol/L氢氧化钠调节pH至7.0,121 ℃高压灭菌15 min备用),36 ℃有氧培养6 h,以生理盐水调整浓度至0.5麦氏浓度,作为受试菌初始菌液,按10:1浓度加入试验体系。
(3)样本药液的处理
准确称取生山楂,焦山楂和类黑素浸膏各10 g,加入100 ml去离子水,超声振荡处理,期间手动震摇数次,直至样本完全溶解,配制10%母液,并经115 ℃高压灭菌处理15 min后4 ℃保存备用。
(4)乙酸含量测定
①样本前处理:将经过微生物培养的溶液1 ml,经过高速离心机4 000 r/min离心,之后再过0.2 µm有机相滤头于进样瓶,样品量大于0.5 ml,或者使用内插管,上机测定。
②标准溶液及标准曲线:称取60.05 g乙酸于100 ml容量瓶,用一级水定容至刻度,摇匀,作为储备标准溶液,浓度为101.33 mmol/L。将标准储备溶液依次稀释1、3、10、20、100、200倍得标准工作溶液。
③色谱条件:洗针液为甲醇,进样量0.5 µl,进样口温度240 ℃;压力6.1219 psi;分流比10:1,流量为1.0 ml;升温程序:初始温度:100 ℃,保持0 min;梯度一:以5 ℃/min升到120 ℃,保持0 min;梯度二:以20 ℃/min升到200 ℃,保持10 min;总运行时间:18 min;检测器(FID)温度:240 ℃;空气流量:300 ml/min;氢气流量:33 ml/min;尾吹氮气流量:20 ml/min;数据采集频率/峰宽:20 Hz/0.01 min。
1.3 统计学方法
使用SPSS 22.0进行独立样本t检验,数据以平均数±标准差(
$\bar x \pm s$ )表示,P<0.05认为存在显著性差异。2. 结果
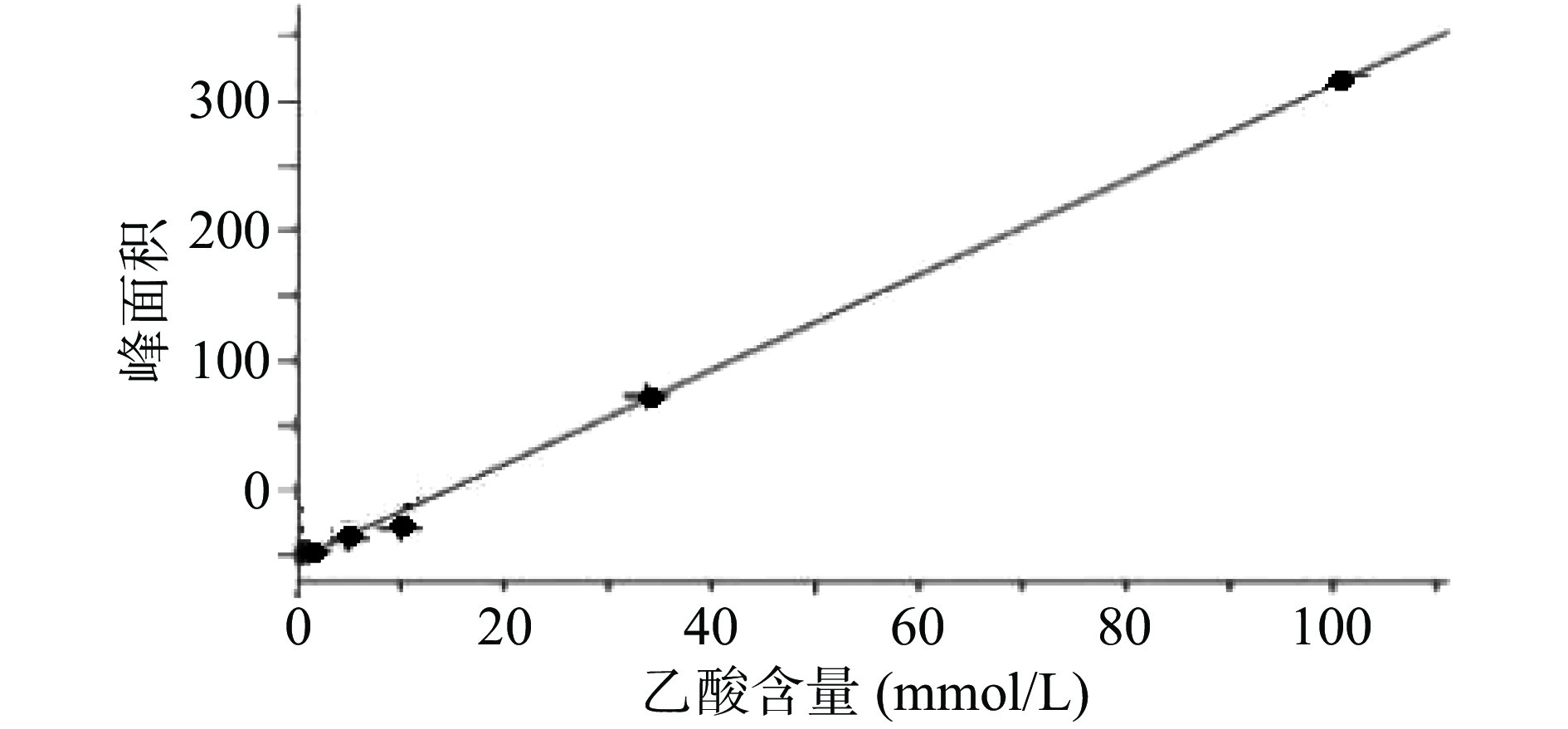
2.1 乙酸标准曲线的建立
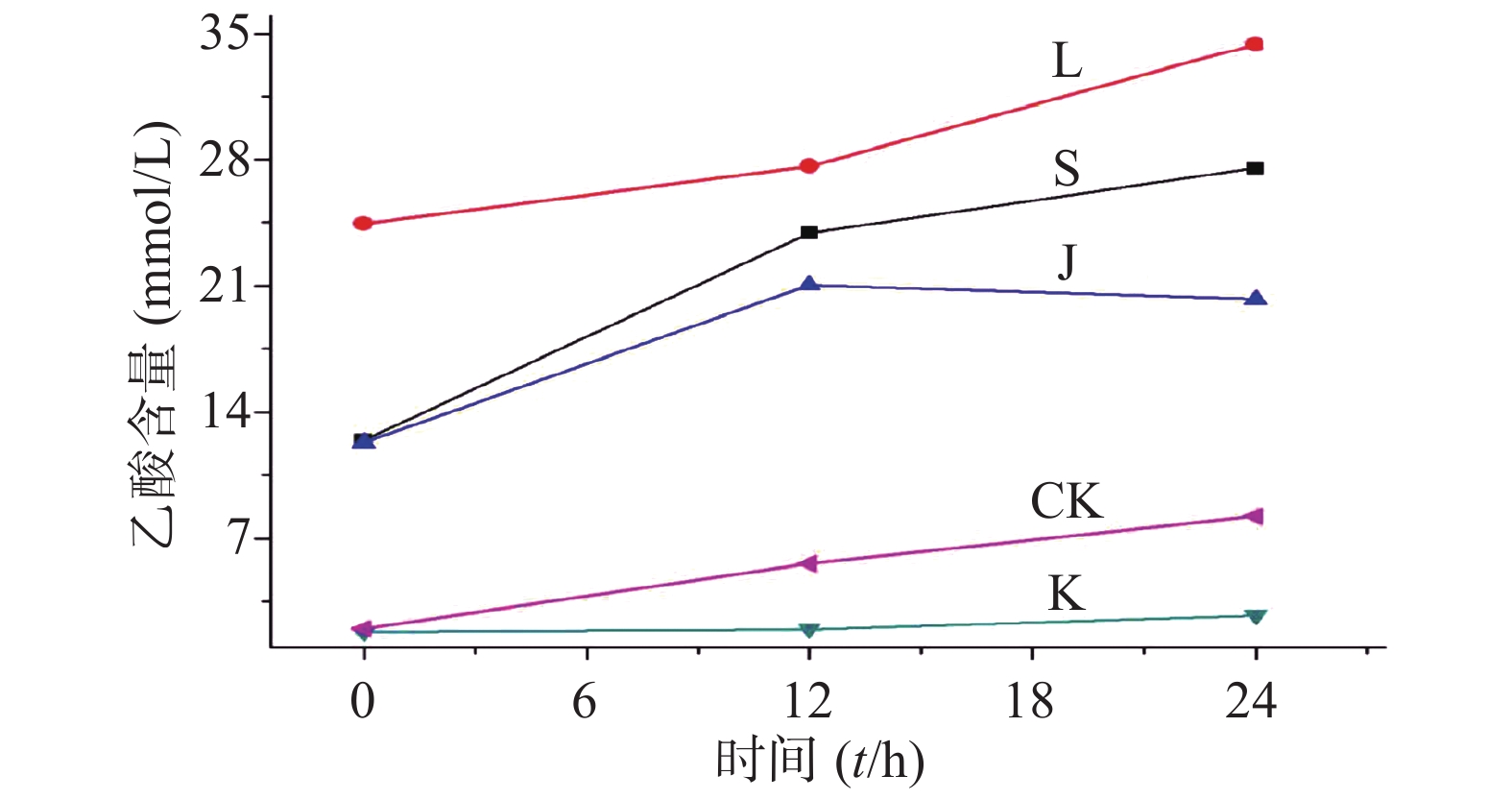
乙酸浓度在0.51~101.33 mmol/L线性关系良好。以乙酸峰面积(Y)为纵坐标,乙酸含量(X)为横坐标,绘制标准曲线,得到线性回归方程为Y=3.670 5X−4.300 8,r=0.999 0,残留标准误差为6.644 2,如图1所示。
2.2 山楂饮片及类黑素对双歧杆菌体外生长的影响
生山楂和焦山楂加速生长期双歧杆菌的生长繁殖,达稳定期后,由于生山楂中多种物质被分解,菌群产生大量代谢废物,于衰亡期加速双歧杆菌的衰亡;由于焦山楂中多种物质被分解,菌群产生大量代谢废物,于衰亡期加速双歧杆菌的衰亡;但因焦山楂中存在类黑素且其他物质较少,衰亡速率慢于生山楂组;类黑素加速生长期双歧杆菌的生长繁殖,但由于无其他物质,其生长速率慢于生山楂组,但在衰亡期中明显改变双歧杆菌生长规律,使生长期延长(生长速率变缓),双歧杆菌衰亡延后,如图2。
2.3 山楂饮片及类黑素对大肠杆菌体外生长的影响
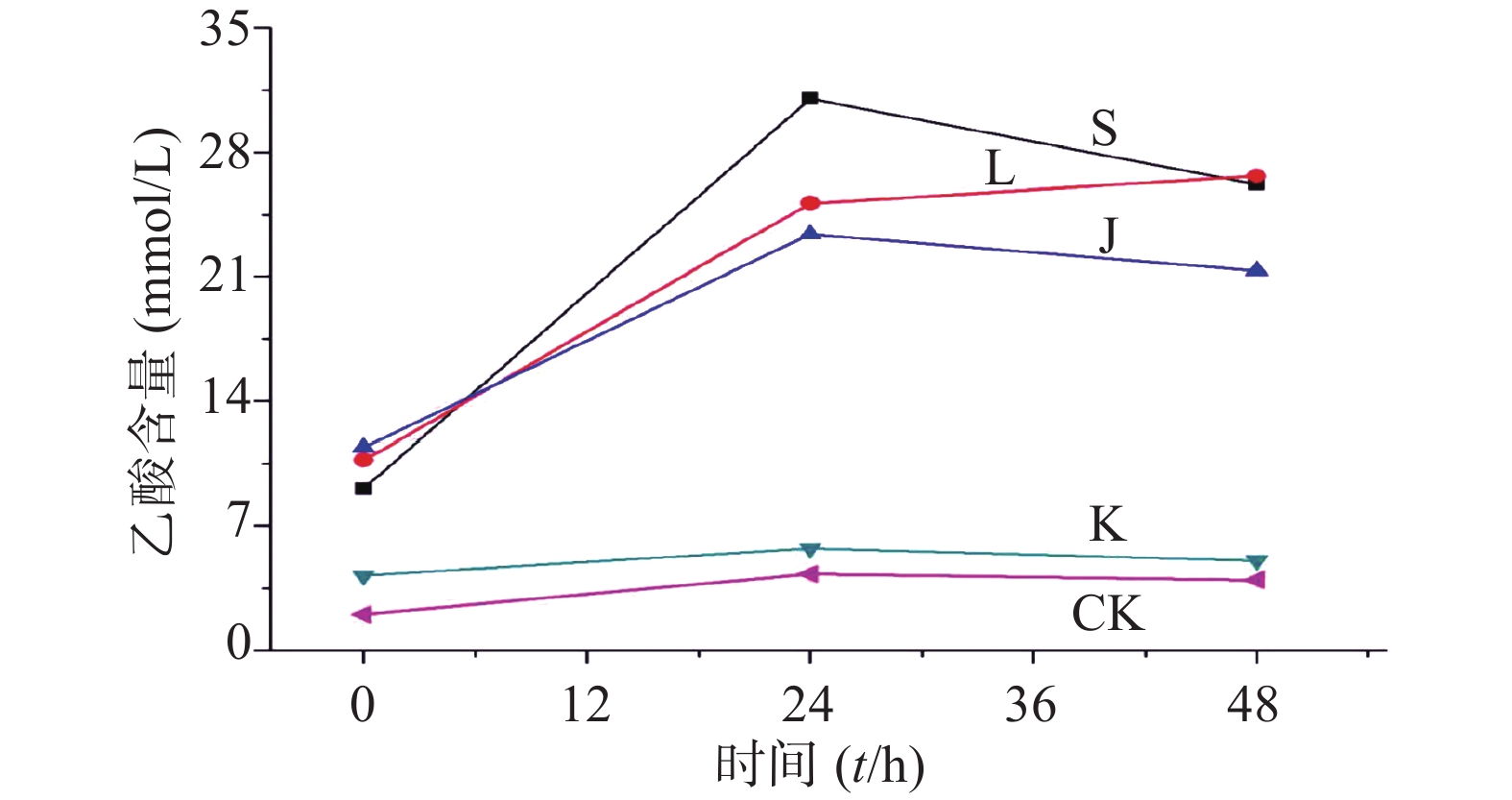
生山楂促进大肠杆菌生长期前期的生长繁殖,但由于代谢废物的逐渐增加,乙酸堆积,使生长速率逐渐变缓;焦山楂促进大肠杆菌生长期前期的生长繁殖,但由于类黑素及代谢废物的影响使生长期变短,稳定期提前;类黑素对大肠杆菌生长期前期无明显影响,但生长期后期明显促进大肠杆菌的生长繁殖,如图3所示。
3. 讨论
3.1 焦山楂炮制工艺研究
《中国药典》一部中对焦山楂炮制方法为:取净山楂,中火条件下炒至药材表面焦褐色,内部焦黄色,并具有焦香气味。因无可控工艺参数,焦山楂炮制过程中易出现饮片表面以及内部颜色不均一,山楂炒制成品质量不稳定等情况。结合课题组前期实验,采用分别100、150、200和250 g净山楂为炮制对象,中火条件为(340~380)℃、(380~420)℃和(420~460)℃,炮制时间为8、10、12和14 min;不同质量同一批号的净山楂在不同的中火条件下炮制不同的时间,采用非接触式红外测温仪检测炒制温度,并以炒锅初温和山楂药材炒制末温辅助控温。实验筛选出150 g净山楂中火条件(380~420)℃下炒制10 min,可得到质量稳定,颜色均一的焦山楂。
3.2 焦山楂类黑素提取工艺研究
类黑素的提取方法主要是水浸提法,Borrelli等[8]在90 ℃条件下,采用1:6料液比,对咖啡中的类黑素进行水提;Langner等[9]在室温条件下采用1:12料液比,水浸提1 h,提取到土豆类黑素粗制品。类黑素成分复杂,提纯困难。目前,主要的纯化方法有大孔树脂、超滤和凝胶层析等方法。何健[10]等发现X-5大孔树脂是曲霉型豆豉类黑素的最佳吸附树脂。秦礼康等[11]利用S-8树脂分离得到豆豉两个类黑素组分。本实验在水浸提法的基础上进行改良,最终获得最优提取工艺。结果显示类黑色素在420 nm处有较强吸收[12]。
3.3 气相色谱条件的筛选
实验采用气相色谱法检测菌群代谢物乙酸的含量。参照文献[13-14],结果显示其色谱条件对于本样品分析效果不佳;在柱温选择中,恒温法对乙酸检测效果不理想,峰形不稳定,因此实验采取梯度升温。经反复试验,最终获得正文中的检测参数,分离效果好,可作为本实验乙酸检测条件。
-
表 1 目标化合物1的体外光动力抗癌活性(IC50,μmol/L)
化合物 B16-F10细胞 暗毒/光毒比 HepG2细胞 暗毒/光毒比 暗毒性 光毒性 暗毒性 光毒性 化合物 1 46.84±8.46*, ΔΔΔ 0.73±0.16**, ΔΔΔ 64.2 50.80±6.45**, #, ΔΔΔ 0.90±0.22**, ΔΔΔ 56.4 二氢卟吩e6 69.72±4.69 3.36±0.59 20.8 70.38±10.9 2.75±0.41 25.6 他拉泊芬 254.8±18.8 11.31±3.88 22.5 176.4±28.4 15.47±5.07 11.4 5-Fu 35.80±6.68 NTa − 39.16±2.7 NTa − NTa:未测定;*P < 0.05,**P < 0.01,与二氢卟吩 e6组比较;#P < 0.05,与5-Fu组比较;ΔΔΔP < 0.001,与他拉泊芬组比较。 -
[1] AGOSTINIS P, BERG K, CENGEL K A, et al. Photodynamic therapy of cancer: an update[J]. CA Cancer J Clin, 2011, 61(4):250-281. doi: 10.3322/caac.20114 [2] ABRAHAMSE H, HAMBLIN M R. New photosensitizers for photodynamic therapy[J]. Biochem J, 2016, 473(4):347-364. doi: 10.1042/BJ20150942 [3] DOUGHERTY T J. An update on photodynamic therapy appli-cations[J]. J Clin Laser Med Surg, 2002, 20(1):3-7. doi: 10.1089/104454702753474931 [4] DROGAT N, GADY C, GRANET R, et al. Design and synthe-sis of water-soluble polyaminated chlorins and bacteriochlorins - with near-infrared absorption[J]. Dyes Pigments, 2013, 98(3):609-614. doi: 10.1016/j.dyepig.2013.03.018 [5] 刘明辉, 刘俊宏, 韩贵焱, 等. 二氢卟吩p6-13, 15-环酰亚胺类光敏剂的设计合成[J]. 药学实践杂志, 2017, 35(1):26-30,35. [6] MENG Z, YU B, HAN G Y, et al. Chlorin p6-based water-soluble amino acid derivatives as potent photosensitizers for photodynamic therapy[J]. J Med Chem, 2016, 59(10):4999-5010. doi: 10.1021/acs.jmedchem.6b00352 [7] ZHANG X J, MENG Z, MA Z Q, et al. Design and synthesis of novel water-soluble amino acid derivatives of chlorin p6 ethers as photosensitizer[J]. Chinese Chem Lett, 2019, 30(1):247-249. doi: 10.1016/j.cclet.2018.04.029 [8] 马福家, 孟志, 张星杰, 等. 二氢卟吩p6醚类光敏剂的合成及光动力抗癌活性研究[J]. 药学实践杂志, 2020, 38(1):52-56. [9] 张丹萍, 陈志龙, 杨晓霞, 等. 光动力药物的研究与开发[J]. 药学进展, 2007, 31(12):529-535. doi: 10.3969/j.issn.1001-5094.2007.12.001 [10] 闵祥燕, 曹宁, 严懿嘉, 等. 光动力新药帕利泊芬研究进展[J]. 药学进展, 2019, 43(3):231-237. [11] TAHMASEBI H, KHOSHGARD K, SAZGARNIA A, et al. Enhancing the efficiency of 5-aminolevulinic acid-mediated photodynamic therapy using 5-fluorouracil on human melanoma cells[J]. Photodiagn Photodyn, 2016, 13:297-302. doi: 10.1016/j.pdpdt.2015.08.011 [12] ZHAO H Y, YIN R, WANG Y, et al. Modulating mitochon-drial morphology enhances antitumor effect of 5-ALA-mediated photodynamic therapy both in vitro and in vivo[J]. J Photoch Photobio B, 2017, 176:81-91. doi: 10.1016/j.jphotobiol.2017.09.017 [13] ZHANG L L, JI Z J, ZHANG J, et al. Photodynamic therapy enhances skin cancer chemotherapy effects through autophagy regulation[J]. Photodiagn Photodyn, 2019, 28:159-165. doi: 10.1016/j.pdpdt.2019.08.023 [14] 姚建忠, 沈卫镝, 陈文晖, 等. 二氢卟吩e6的合成及其光敏化力和肿瘤光生物活性[J]. 中国医药工业杂志, 2000, 31(5):215-217. doi: 10.3969/j.issn.1001-8255.2000.05.009 [15] CHEN H, WARUNA JINADASA R G, JIAO L J, et al. Chlo-rin e6 131: 152-anhydride: a key intermediate in conjugation reactions of chlorin e6[J]. Eur J Org Chem, 2015, 2015(17):3661-3665. doi: 10.1002/ejoc.201500478 -





 下载:
下载:
 下载:
下载: